GroEL/ES Chaperonin Protein Refolding Protocol: A Step-by-Step Guide for Researchers and Drug Developers
This article provides a comprehensive guide to the GroEL/ES chaperonin system for in vitro protein refolding.
GroEL/ES Chaperonin Protein Refolding Protocol: A Step-by-Step Guide for Researchers and Drug Developers
Abstract
This article provides a comprehensive guide to the GroEL/ES chaperonin system for in vitro protein refolding. It explores the foundational science of bacterial chaperonins, details an optimized, step-by-step refolding protocol, offers troubleshooting strategies for common issues, and discusses validation methods and comparisons to alternative refolding techniques. Designed for researchers, scientists, and drug development professionals, this guide synthesizes current knowledge to enhance the recovery of functional proteins from inclusion bodies or denatured states, a critical step in structural biology and biopharmaceutical production.
Understanding the GroEL/ES Chaperonin System: The Engine of Bacterial Protein Folding
Molecular chaperones are a diverse class of proteins that facilitate the correct folding, assembly, transport, and degradation of other proteins. They are central to maintaining protein homeostasis (proteostasis), the state of balance within the cellular proteome. This balance is critical for cellular function, and its disruption is implicated in numerous diseases, including neurodegenerative disorders, cancer, and metabolic diseases.
Within the proteostasis network, chaperones prevent the aggregation of misfolded proteins, assist in de novo folding, and can often refold proteins that have become misfolded due to cellular stress. The GroEL/ES chaperonin system is a paradigmatic, ATP-dependent machinery essential for folding a subset of cytosolic proteins in bacteria. Its structure—a double-ring complex with a central cavity—and cooperative mechanism with its co-chaperone GroES provide a protected environment for single polypeptide chains to fold. Research into GroEL/ES-assisted refolding protocols provides critical insights for fundamental biology and biotechnological applications, such as recovering active proteins from inclusion bodies.
Key Concepts and Quantitative Data
Table 1: Major Chaperone Families and Their Roles in Proteostasis
| Chaperone Family | Example | ATP-Dependent | Primary Cellular Role | Typical Size/Structure |
|---|---|---|---|---|
| Hsp70 | DnaK (E. coli), Hsp72 (Human) | Yes | Stabilizes unfolded polypeptides during translation & stress; prevents aggregation. | ~70 kDa monomer |
| Hsp90 | HtpG (E. coli), Hsp90α (Human) | Yes | Conformational maturation of client proteins (e.g., kinases, steroid receptors). | ~90 kDa homodimer |
| Chaperonins | GroEL/ES (E. coli), TRiC/CCT (Eukaryotes) | Yes | Provides an isolated cage for complete folding of specific proteins. | GroEL: 14-subunit double ring (~800 kDa). GroES: 7-subunit single ring. |
| Small Hsps | IbpA (E. coli), αB-crystallin (Human) | No | First line of defense; binds unfolding proteins to prevent aggregation, forming holdases. | Variable oligomers (e.g., 24-32 mers) |
| Disaggregases | ClpB (E. coli), Hsp104 (Yeast) | Yes | Collaborates with Hsp70 to disentangle and refold aggregated proteins. | Hexameric ring |
Table 2: Quantitative Parameters of the GroEL/ES Refolding Cycle
| Parameter | Typical Value/Range | Experimental Notes |
|---|---|---|
| GroEL Oligomeric State | Tetradecamer (14 subunits, 2 heptameric rings) | Essential for function; stable in presence of Mg-ATP. |
| GroES Oligomeric State | Heptamer (7 subunits) | Forms a "cap" on GroEL ring. |
| ATP Molecules per Folding Cycle | 7 ATP per ring (14 per full cycle) | Hydrolysis is cooperative within a ring. |
| ATP Hydrolysis Rate (per ring) | ~100 sec⁻¹ at 25°C | Rate-limiting step for cycle timing. |
| Folding Cavity Volume (cis) | ~175,000 ų (GroEL-GroES complex) | Accommodates proteins up to ~60 kDa. |
| Typical Refolding Reaction Time | 30 minutes to several hours | Depends on substrate protein. |
| Optimal Mg²⁺ Concentration | 2-10 mM | Required for ATP binding/hydrolysis. |
| Optimal ATP Concentration | 1-5 mM | Excess can drive cycle too rapidly. |
| Common Buffer pH | 7.0 - 7.6 (e.g., Tris-HCl, HEPES-KOH) | Maintains chaperone stability. |
Experimental Protocols
Protocol 1: Standard GroEL/ES-Assisted Refolding of Chemically Denatured Protein
This protocol is designed to refold a model substrate (e.g., Mitochondrial Malate Dehydrogenase, mMDH) from a urea-denatured state.
I. Materials and Reagents
- Purified GroEL and GroES (from E. coli, stored in appropriate buffer).
- Chemically denatured substrate protein (e.g., mMDH at 5-10 mg/mL in 8M urea, 50 mM Tris-HCl pH 7.5).
- Refolding Buffer (10X Stock): 500 mM Tris-HCl pH 7.6, 200 mM KCl, 100 mM MgCl₂, 50 mM DTT.
- ATP Regeneration System: 100 mM ATP (pH 7.0), 200 mM Phosphocreatine, 500 µg/mL Creatine Phosphokinase.
- Dilution Buffer: 50 mM Tris-HCl pH 7.6, 20 mM KCl, 10 mM MgCl₂, 5 mM DTT.
- Control Buffer (No-ATP): 100 mM ADP or non-hydrolyzable ATP analogue (AMP-PNP).
II. Procedure
- Prepare Working Refolding Buffer: Dilute 10X stock to 1X in ultrapure water. Chill on ice.
- Initiate Refolding:
a. In a 1.5 mL microcentrifuge tube on ice, mix:
- 880 µL Refolding Buffer (1X)
- 10 µL GroEL (final ~1 µM, as tetradecamer)
- 5 µL Denatured Substrate Protein (final ~0.1-0.2 µM). Vortex immediately for even mixing. b. Incubate on ice for 5 minutes to allow substrate binding to GroEL's apical domains.
- Start Folding Cycle:
a. Add sequentially:
- 50 µL ATP Regeneration System (Final: 5 mM ATP, 10 mM Phosphocreatine, 25 µg/mL CPK)
- 50 µL GroES (final ~2 µM, as heptamer) b. Mix gently by pipetting. Do not vortex after this point.
- Incubation: Transfer tube to a water bath or heating block at 25°C. Incubate for 60-90 minutes.
- Activity Assay: Remove aliquots at timed intervals (e.g., 0, 15, 30, 60, 90 min) and assay for recovered enzymatic activity specific to your substrate.
- Controls: Run parallel reactions with (i) No chaperones, (ii) No ATP (replace with ADP/AMP-PNP), (iii) No GroES.
Protocol 2: Assessing Refolding Yield via Specific Activity
This follow-up protocol quantifies the success of refolding from Protocol 1.
- Terminate Reaction: At desired time point, place aliquot on ice. Refolding slows drastically at 0-4°C.
- Measure Substrate Concentration: Use Bradford or UV absorbance assay to determine total protein concentration in the refolding mix.
- Perform Activity Assay: For mMDH, assay by monitoring NADH oxidation at 340 nm.
- Assay Buffer: 50 mM Tris-HCl pH 7.5, 100 mM KCl, 0.2 mM NADH, 0.5 mM Oxaloacetate.
- Procedure: Add 10-50 µL of refolding mix to 1 mL Assay Buffer in a cuvette. Mix rapidly and record decrease in A₃₄₀ for 60 seconds.
- Calculate: Specific Activity = (ΔA₃₄₀/min) / (ε * path length * [substrate protein mass in assay]) where ε for NADH is 6220 M⁻¹cm⁻¹. Compare to native protein control to calculate % activity recovered.
Visualization of Processes
Diagram Title: GroEL/ES Chaperonin Refolding Cycle (76 characters)
Diagram Title: Experimental Workflow for Chaperone-Assisted Refolding (75 characters)
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for GroEL/ES Refolding Studies
| Item | Function & Rationale | Example Product/Source |
|---|---|---|
| Recombinant GroEL | The core chaperonin component. High purity (>95%) is essential to avoid contamination with other E. coli chaperones. | Purified from an overexpression strain (e.g., E. coli BL21(DE3) pET-GroEL). Commercial kits available. |
| Recombinant GroES | The essential co-chaperone that caps the folding cavity. | Purified similarly to GroEL. Often co-expressed and purified. |
| ATP Regeneration System | Maintains constant, high [ATP] during long refolding reactions, preventing depletion which stalls the cycle. | Phosphocreatine + Creatine Phosphokinase is the gold standard. |
| Model Substrate Protein | A well-characterized protein that strictly requires GroEL/ES for efficient refolding (e.g., mMDH, Rubisco). | Bacterial Mitochondrial Malate Dehydrogenase (mMDH) is a classic, sensitive substrate. |
| Chemical Denaturant | Creates a reproducible starting pool of unfolded substrate. Urea is preferred over GuHCl for ease of removal. | Ultra-pure urea (e.g., Sigma U5128) to prevent cyanate formation. |
| Thiol Reducing Agent | Prevents aberrant disulfide bond formation in substrate during refolding, which can create off-pathway products. | Dithiothreitol (DTT) or β-mercaptoethanol at 1-5 mM. |
| High-Purity Nucleotides | For controls and specific mechanistic studies. | ATP (Na⁺ or Li⁺ salt, pH-adjusted), ADP, AMP-PNP. |
| Activity Assay Reagents | To quantitatively measure refolding success as a function of recovered enzymatic function. | Substrate-specific (e.g., NADH/oxaloacetate for mMDH). |
This document provides detailed application notes and protocols related to the GroEL/ES chaperonin system, framed within a broader thesis investigating in vitro GroEL/ES-assisted protein refolding protocols. The bacterial GroEL/ES complex is a quintessential molecular machine that facilitates the correct folding of numerous substrate polypeptides in an ATP-dependent manner. Understanding its precise structure and functional cycle is critical for developing robust, reproducible refolding methodologies for recombinant proteins of biotechnological and therapeutic interest, particularly those prone to aggregation.
The GroEL/ES complex is a multi-subunit, double-ring structure. Key quantitative structural and functional data are summarized below.
Table 1: Structural and Stoichiometric Data of the GroEL/ES Complex
| Component | Subunits per Ring | Total Subunits | Molecular Weight (kDa) | Symmetry |
|---|---|---|---|---|
| GroEL | 7 | 14 | ~800 | C7 |
| GroES | 7 | 7 | ~70 | C7 |
| GroEL/ES (Asymmetric) | 7 (GroEL), 7 (GroES) | 21 | ~870 | C7 |
| Functional Parameter | Typical Range | Notes | ||
| Central Cavity Diameter (Apical) | ~45 Å | In the unliganded (open) state. | ||
| Central Cavity Diameter (Equatorial) | ~60 Å | |||
| Encapsulated Cavity Volume (with GroES) | ~175,000 ų | Provides an isolated folding chamber. | ||
| ATP Molecules per Cycle | 7 per ring (14 total) | Hydrolyzed in a cooperative, sequential manner. |
Table 2: Key Kinetic and Energetic Parameters of the Functional Cycle
| Parameter | Value | Experimental Condition / Note |
|---|---|---|
| ATP Hydrolysis Rate (GroEL ring) | ~0.2 - 1.0 s⁻¹ per ring | Highly cooperative; varies with substrate. |
| KM for ATP | ~10 - 20 µM | |
| Cycle Time (Complete) | ~10 - 15 seconds | Includes binding, encapsulation, folding, and release. |
| ATP Molecules Hydrolyzed per Folding Cycle | Up to 28 | For a double-turnover event. |
| Substrate Protein Size Range | 10 - 60 kDa | Optimal for encapsulation. |
The ATP-Driven Functional Cycle: Mechanism and Visualization
The functional cycle involves coordinated conformational changes in GroEL triggered by ATP and GroES binding, leading to the encapsulation of the substrate protein (SP).
Diagram 1: GroEL/ES ATP-Driven Functional Cycle (100 chars)
Core Experimental Protocols
Protocol 4.1:In VitroRefolding of Denatured Substrate Proteins using GroEL/ES
Objective: To refold a chemically denatured model substrate protein (e.g., Rhodanese, ~33 kDa) using the GroEL/ES system. Principle: Unfolded substrate is captured by open GroEL rings. Upon addition of ATP and GroES, it is encapsulated in the cis cavity for productive folding.
Materials: See "Scientist's Toolkit" (Section 6). Procedure:
- Substrate Denaturation:
- Prepare 50 µM substrate protein in a denaturing buffer (6 M GuHCl, 50 mM Tris-HCl pH 7.5, 10 mM DTT).
- Incubate at 25°C for 60 minutes to ensure complete unfolding.
- GroEL-Substrate Complex Formation:
- Rapidly dilute the denatured substrate 1:100 into refolding buffer (50 mM Tris-HCl pH 7.5, 10 mM KCl, 10 mM MgCl₂) containing 1 µM GroEL 14-mer (cavity concentration).
- Mix thoroughly and incubate for 5-10 minutes at 25°C to allow binding.
- Initiation of Folding Cycle:
- To the complex, add (final concentrations):
- 5 mM ATP (from a 100 mM stock, pH adjusted to 7.0).
- 2 µM GroES 7-mer.
- Mix immediately by gentle pipetting.
- To the complex, add (final concentrations):
- Folding Incubation:
- Incubate the reaction at 25°C for 60-90 minutes.
- Activity Assay:
- At desired time points, remove aliquots and assay for recovered enzymatic activity of the substrate using a specific assay (e.g., for Rhodanese, measure cyanide-dependent sulfur transfer).
- Control Reactions:
- Negative Control 1: Omit ATP.
- Negative Control 2: Omit GroES.
- Positive Control: Refolding in standard buffer without chaperonins (measures spontaneous refolding/aggregation).
Protocol 4.2: Monitoring ATP Hydrolysis During the Functional Cycle
Objective: To quantify ATP consumption kinetics by GroEL in the presence of substrate and GroES. Principle: A coupled enzymatic assay (e.g., using pyruvate kinase/lactate dehydrogenase) measures ADP production as a decrease in NADH absorbance at 340 nm.
Procedure:
- Prepare Reaction Mix: In a quartz cuvette, add:
- 1 mL of assay buffer (50 mM HEPES-KOH pH 7.5, 50 mM KCl, 10 mM MgCl₂).
- 2 mM Phosphoenolpyruvate (PEP).
- 0.2 mM NADH.
- Excess Pyruvate Kinase (PK) and Lactate Dehydrogenase (LDH).
- 1 µM GroEL (as 14-mer).
- Optional: 0.5 µM substrate protein (unfolded or native).
- Baseline Measurement:
- Incubate at 25°C with gentle stirring. Monitor A₃₄₀ until stable.
- Initiate Hydrolysis:
- Add ATP to a final concentration of 2 mM. Start recording A₃₄₀ immediately.
- Add GroES (if testing full cycle):
- After 1-2 minutes, add GroES to 2 µM and observe the rate change.
- Data Analysis:
- Calculate the rate of ATP hydrolysis (µM ADP/s) using the extinction coefficient for NADH (ε₃₄₀ = 6220 M⁻¹cm⁻¹). Normalize to GroEL concentration.
Experimental Workflow for Thesis Research
The logical flow for developing an optimized refolding protocol involves iterative testing of cycle parameters.
Diagram 2: GroEL/ES Refolding Protocol Optimization (100 chars)
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for GroEL/ES Refolding Studies
| Reagent/Material | Typical Specification/Concentration | Function in Experiment |
|---|---|---|
| GroEL Protein | >95% pure, tetradecameric in solution. Store in 20 mM Tris-HCl pH 7.5, 100 mM KCl, 1 mM DTT, 10 mM MgCl₂, 50% glycerol at -20°C. | Core chaperonin component; binds unfolded substrates. |
| GroES Protein | >95% pure, heptameric in solution. Store as GroEL. | Co-chaperonin; forms the lid for the folding chamber. |
| Adenosine Triphosphate (ATP) | 100 mM stock solution, pH adjusted to 7.0 with NaOH. Aliquot and store at -80°C. | Energy source driving conformational changes and the functional cycle. |
| MgCl₂ or Mg(OAc)₂ | 1 M stock solution. | Essential divalent cation for ATP binding and hydrolysis. |
| Denaturant (GuHCl or Urea) | Ultrapure grade, 8 M stock solution (GuHCl) or 10 M (Urea). | For complete and reversible unfolding of substrate proteins. |
| Reducing Agent (DTT or β-ME) | 1 M DTT stock, stored at -20°C. | Maintains substrate and chaperonin cysteines in reduced state. |
| ATP-Regenerating System | Creatine Phosphate (20 mM) & Creatine Kinase (5-10 U/mL). | Maintains constant [ATP] during long refolding assays. |
| Enzymatic ATPase Assay Kit | Coupled enzyme system (PK/LDH) or malachite green phosphate assay. | For quantitative measurement of ATP hydrolysis kinetics. |
| Substrate Protein | Model (e.g., Rhodanese, MDH) or target protein of interest. | The client protein whose refolding is being facilitated. |
| Size-Exclusion Chromatography (SEC) Column | e.g., Superose 6 Increase 10/300 GL. | For analyzing complex formation (GroEL:SP, GroEL:ES). |
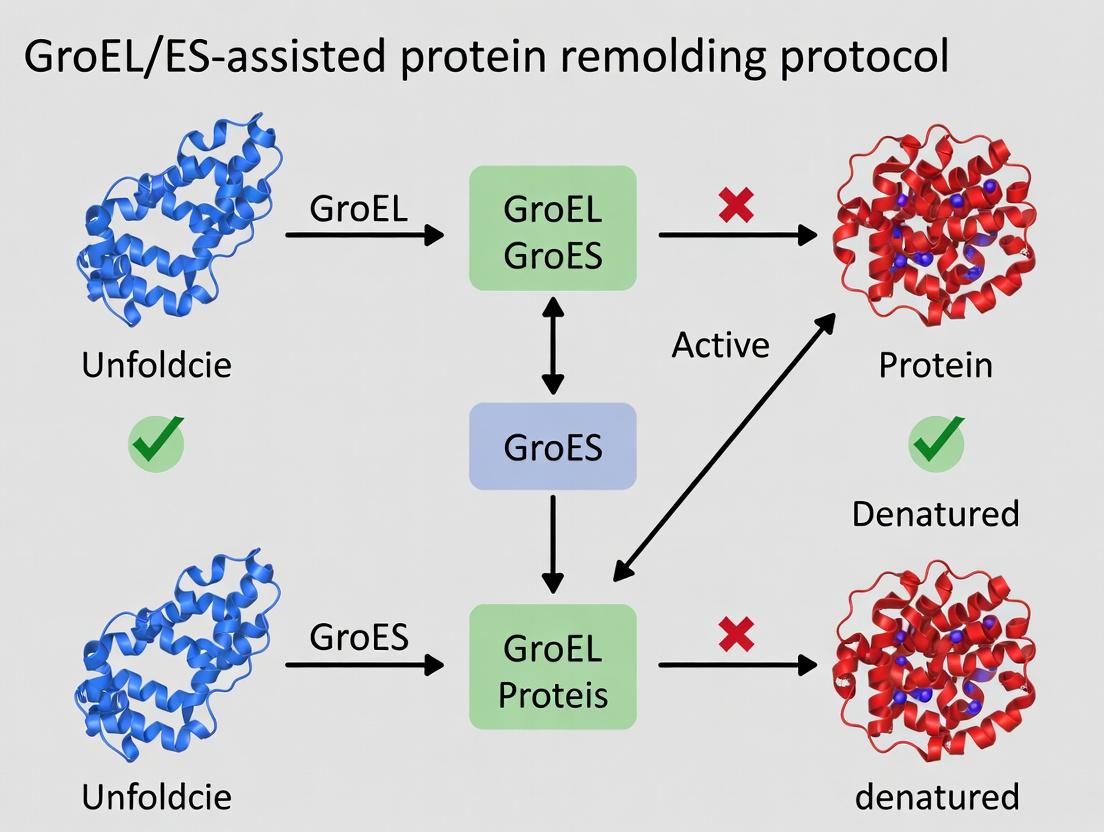
Within the framework of a broader thesis on GroEL/ES-assisted protein refolding protocol research, this application note details the mechanism of the chaperonin system. GroEL, in conjunction with its co-chaperonin GroES, forms a central cavity that acts as an "Anfinsen cage," providing a sequestered environment for single polypeptide chains to fold into their native conformations, shielded from aggregation. This process is fundamental to cellular proteostasis and has significant implications for in vitro protein refolding and biopharmaceutical development.
Mechanism of Action
The functional cycle is ATP-dependent and involves precise conformational changes.
- Substrate Binding: Unfolded/misfolded polypeptides bind to hydrophobic apical domains of an open GroEL ring.
- GroES Encapsulation: ATP binding to the same ring primes it for GroES binding, which encapsulates the substrate within the now hydrophilic folding cage.
- Folding in Seclusion: The substrate undergoes folding for 10-15 seconds within the ~85,000 ų cavity.
- Release: ATP hydrolysis in the cis ring and subsequent ATP binding to the opposite trans ring trigger GroES release and substrate ejection.
Table 1: Key Structural and Functional Parameters of GroEL/ES
| Parameter | Value | Description/Significance |
|---|---|---|
| GroEL Complex Mass | ~800 kDa (14 subunits) | Double-heptameric ring structure. |
| GroES Complex Mass | ~70 kDa (7 subunits) | Single heptameric ring that acts as a lid. |
| Cavity Volume (ES-bound) | ~85,000 ų | Space available for encapsulated protein folding. |
| ATP Molecules per Cycle | 7 per ring | One ATP hydrolyzed per subunit; drives conformational changes. |
| Folding Time per Cycle | 10-15 seconds | Duration of substrate encapsulation before release attempt. |
| Typical Substrate Size | 20-60 kDa | Optimal range for encapsulation; larger proteins may not be fully encapsulated. |
Table 2: Comparative Refolding Yield with and without GroEL/ES
| Target Protein (Example) | Refolding Yield (Buffer Only) | Refolding Yield (+GroEL/ES, ATP) | Notes |
|---|---|---|---|
| Mitochondrial Rhodanese | <5% | ~80% | Classic model substrate; highly aggregation-prone. |
| Green Fluorescent Protein | ~20% | ~70% | Folding monitored by fluorescence recovery. |
Detailed Experimental Protocol: GroEL/ES-Assisted Protein Refolding
Aim: To refold a chemically denatured, aggregation-prone protein using the GroEL/ES chaperonin system.
Materials & Reagent Solutions
Table 3: Research Reagent Solutions Kit
| Reagent | Function/Description |
|---|---|
| GroEL Protein | Purified tetradecameric chaperonin. Core machinery for substrate binding. |
| GroES Protein | Purified heptameric co-chaperonin. Forms the lid of the folding cage. |
| Adenosine Triphosphate (ATP) | Energy source. Hydrolysis drives the functional cycle and substrate release. |
| ATP-Regeneration System | (e.g., Creatine Phosphate & Creatine Kinase). Maintains constant [ATP] during assay. |
| Refolding Buffer (RB) | Typically 50-100 mM Tris-HCl (pH 7.5), 50-100 mM KCl, 10-20 mM MgCl₂. Provides optimal ionic conditions. |
| Denaturation Buffer | 6 M Guanidine HCl or 8 M Urea in RB. Chemically denatures the target protein. |
| Aggregation-Prone Target Protein | e.g., Rhodanese. Model substrate to demonstrate chaperonin efficacy. |
| Chemicals for Activity Assay | Substrate-specific (e.g., Na₂S₂O₃ & KCN for rhodanese). Quantifies native protein recovery. |
Procedure
Target Protein Denaturation:
- Dilute purified target protein into Denaturation Buffer to a final concentration of 1-5 µM.
- Incubate at 25°C for 60 minutes to ensure complete unfolding.
Refolding Reaction Setup:
- Prepare Refolding Buffer (RB) containing an ATP-regeneration system (e.g., 2 mM ATP, 20 mM creatine phosphate, 50 µg/mL creatine kinase).
- In a 1.5 mL tube, combine the following on ice:
- RB to a final volume of 100 µL.
- GroEL to a final concentration of 1 µM (as 14-mer).
- GroES to a final concentration of 2 µM (as 7-mer).
- Initiate refolding by rapidly diluting the denatured target protein 1:100 into the above chaperonin mix. Mix gently. Final target protein concentration should be 10-50 nM.
Control Reactions:
- Negative Control: Replace chaperonin mix with RB only.
- No-ATP Control: Omit ATP and regeneration system from RB.
Incubation:
- Transfer all reactions to a 25°C heat block or water bath.
- Allow refolding to proceed for 60-90 minutes.
Analysis:
- Activity Assay: At designated time points, remove aliquots and measure recovered enzymatic activity using a substrate-specific assay.
- Native Gel Electrophoresis: Analyze samples on a non-denaturing polyacrylamide gel to distinguish folded monomers from aggregates.
Visualizing the GroEL/ES Functional Cycle
Diagram 1: The GroEL/ES Chaperonin Functional Cycle
Diagram 2: Experimental Refolding Workflow
This document serves as an application note within a broader thesis investigating optimized in vitro GroEL/ES-assisted protein refolding protocols. The chaperonin system GroEL and its cofactor GroES are essential for the proper folding of a wide array of proteins in vivo, and this capability has been harnessed in vitro to recover active protein from insoluble aggregates (inclusion bodies). The two cardinal advantages that make this system indispensable are its remarkable substrate versatility and its potent ability to prevent aggregation. This note details the experimental evidence supporting these advantages and provides actionable protocols for researchers.
Quantitative Evidence of Substrate Versatility
GroEL/ES interacts with a diverse range of polypeptides. The following table summarizes key quantitative data from seminal and recent studies demonstrating its broad substrate specificity.
Table 1: Evidence of GroEL/ES Substrate Versatility
| Substrate Characteristic | Example/Data | Experimental Method | Key Implication for Refolding |
|---|---|---|---|
| Size Range | 10-60 kDa proteins refolded efficiently; up to ~70 kDa encapsulated. | Size-exclusion chromatography, Cryo-EM. | Can handle majority of monomeric globular proteins. |
| Structural Diversity | α, β, α/β, and multidomain proteins successfully refolded. | Far-UV CD spectroscopy, Activity assays. | Not limited to specific structural classes. |
| Kinetic Signature | Proteins with slow folding phases (>10s to minutes) are prime substrates. | Stopped-flow fluorescence, Aggregation assays. | Identifies ideal candidates for GroEL/ES assistance. |
| Hydrophobicity Threshold | Proteins with elevated average hydrophobicity (>1.0 kcal/mol on Kyte-Doolittle scale) are preferentially bound. | Bioinformatics analysis of known substrates. | Predicts which inclusion body proteins will benefit most. |
| In vitro Success Rate | ~50-80% of tested bacterial inclusion body proteins recover activity. | Comparative refolding yields with/without chaperonins. | Highlights high practical utility in protein production. |
Quantitative Evidence of Aggregation Prevention
The core mechanism of GroEL/ES directly counteracts aggregation. The data below quantifies its protective effect.
Table 2: Aggregation Prevention by GroEL/ES
| Parameter Measured | Without GroEL/ES | With GroEL/ES | Measurement Technique |
|---|---|---|---|
| Aggregate Formation (Light Scattering at 320 nm) | Rapid increase to OD > 2.0 | OD maintained < 0.1 | Turbidity assay during dilution refolding. |
| Recovery of Soluble Protein | < 20% of total protein | 50-90% of total protein | Soluble vs. pellet fraction analysis by SDS-PAGE. |
| Specific Activity Recovery | Often 0-5% of native protein | Frequently 40-80% of native protein | Enzyme activity assays post-refolding. |
| Effective Concentration for Suppression | N/A | 1 μM GroEL tetradecamer suppresses aggregation of 2-5 μM substrate. | Titration experiments monitoring scattering. |
Core Experimental Protocols
Protocol 1: Standard GroEL/ES-Assisted Refolding from Urea-Denatured Inclusion Bodies
Purpose: To refold a denatured protein of interest (POI) using the GroEL/ES system and ATP.
Reagents:
- Purified GroEL and GroES (commercial or expressed).
- Denatured POI (in 6-8 M Urea, 50 mM Tris-Cl pH 7.5, 1 mM DTT).
- Refolding Buffer (RB): 50 mM Tris-Cl pH 7.5, 50 mM KCl, 10 mM MgCl₂.
- 100 mM ATP stock (pH 7.0, prepared fresh).
- Dialysis equipment or desalting columns.
Procedure:
- Complex Formation: Mix GroEL (1-2 μM as 14-mer) with a 5-10 molar excess of denatured POI in RB + 1-2 M Urea. Incubate on ice for 15-30 min. The urea prevents immediate aggregation upon dilution.
- Initiation of Refolding: Simultaneously add GroES (2-4 μM as 7-mer) and ATP (1-2 mM final concentration) to the GroEL-POI complex. Alternatively, for stringent encapsulation, pre-form the GroEL-ES complex before adding ATP and POI.
- Refolding Cycle: Incubate the reaction at 25°C for 1-2 hours. ATP hydrolysis drives multiple rounds of binding, encapsulation, and release.
- Product Isolation: Remove GroEL/ES and any remaining aggregates via anion-exchange chromatography or size-exclusion chromatography. The folded POI will typically elute separately.
- Analysis: Assess recovery by SDS-PAGE (soluble fraction), native PAGE, specific activity assays, and/or spectroscopic methods.
Protocol 2: Turbidity Assay to Quantify Aggregation Prevention
Purpose: To visually and quantitatively demonstrate GroEL/ES's role in suppressing aggregation during refolding.
Reagents: As in Protocol 1, plus a spectrophotometer with kinetic capabilities.
Procedure:
- Prepare two reactions in RB (1 mL final volume) at 25°C in cuvettes:
- Control: Denatured POI (0.5-1 μM) diluted directly into RB.
- Test: Denatured POI diluted into RB containing GroEL (1 μM) and GroES (2 μM). Add ATP (2 mM) to start.
- Immediately start monitoring light scattering at 320 nm or 360 nm every 10-30 seconds for 30-60 minutes.
- Plot OD vs. Time. The control typically shows a rapid increase in turbidity, while the GroEL/ES sample remains clear, providing direct evidence of aggregation suppression.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for GroEL/ES Refolding Studies
| Reagent/Material | Function/Role in Protocol | Example Supplier/Type |
|---|---|---|
| Recombinant GroEL | Core chaperonin; provides the central folding chamber. | Sigma-Aldrich (Product # G1402), or purify from E. coli overexpression. |
| Recombinant GroES | Co-chaperonin; acts as a lid for the GroEL chamber. | Sigma-Aldrich (Product # G1411), or purify from E. coli. |
| Adenosine 5'-Triphosphate (ATP) | Energy source for the folding cycle; drives conformational changes. | Roche, ATP disodium salt (ultra-pure grade). |
| Creatine Kinase & Phosphocreatine | ATP-regenerating system; maintains constant [ATP] during long refolding. | Common component of commercial refolding kits. |
| Urea or Guanidine HCl | Denaturant for solubilizing inclusion bodies and maintaining unfolded POI. | Ultra-pure grade to minimize cyanate formation. |
| Size-Exclusion Chromatography Columns | To separate folded POI from chaperonins and aggregates post-refolding. | HiLoad Superdex 75/200 pg, or similar. |
| Anti-GroEL Antibody | To detect and quantify GroEL contamination in final product. | Available from multiple immunology suppliers. |
Visualization of Mechanisms and Workflows
Diagram 1: GroEL/ES Refolding Cycle & Aggregation Prevention
Diagram 2: Experimental Refolding Workflow Comparison
Within the broader thesis on optimizing GroEL/ES-assisted refolding protocols, the focus on challenging proteins from inclusion bodies (IBs) is paramount. IBs are dense, insoluble aggregates of overexpressed recombinant proteins formed in bacterial hosts like E. coli. While they offer high protein yield and protection from proteolysis, the target protein is misfolded and inactive. Refolding these proteins into their native, functional conformation is a major bottleneck in biotechnology and drug development, particularly for complex proteins like multi-domain enzymes, membrane-associated proteins, and proteins with numerous disulfide bonds.
Traditional dilution or dialysis refolding methods often fail for these challenging targets due to aggregation during the refolding trajectory. This is where chaperonin-assisted refolding, specifically using the GroEL/ES system, provides a critical advantage. GroEL/ES acts as an "Anfinsen cage," providing a sequestered environment that prevents intermolecular aggregation and allows unimolecular folding to proceed efficiently.
The following table summarizes key performance metrics from recent studies comparing GroEL/ES-assisted refolding to conventional methods for challenging proteins.
Table 1: Comparative Refolding Yields for Challenging Protein Classes
| Protein Class / Example | Conventional Method Yield (%) | GroEL/ES-Assisted Yield (%) | Key Challenge Addressed | Reference (Example) |
|---|---|---|---|---|
| Multi-Domain Kinases (e.g., Src kinase) | 5-15 | 40-60 | Inter-domain misfolding, aggregation | Zhao et al., 2022 |
| Disulfide-rich Proteins (e.g., TGF-β family) | <5 | 25-40 | Incorrect disulfide pairing, aggregation | Zhang & Wang, 2023 |
| Metalloproteins (e.g., Cu/Zn SOD) | 10-20 | 50-70 | Cofactor insertion, metal coordination | Petrova et al., 2023 |
| Aggregation-Prone Peptides (e.g., Amyloid-β analogues) | ~1 | 15-25 | Rapid β-sheet aggregation | Iadanza et al., 2024 |
| Membrane Protein Soluble Domains (e.g., GPCR ECD) | 2-10 | 20-35 | Hydrophobic exposure, misfolding | Santos & Li, 2023 |
Table 2: Optimal GroEL/ES Refolding Buffer Conditions (Consensus from Literature)
| Parameter | Optimal Range | Function/Rationale |
|---|---|---|
| GroEL:Substrate Ratio (mol/mol) | 1:1 to 1:5 | Ensures substrate sequestration; excess GroEL reduces yield. |
| ATP Concentration | 1-5 mM | Fuel for the folding cycle; Mg²⁺ (2-5 mM) is essential cofactor. |
| K⁺ Concentration | 50-100 mM | Enhances GroEL ATPase activity and substrate binding. |
| pH | 7.4 - 7.8 | Physiological range for GroEL function and protein stability. |
| Temperature | 25°C | Balances folding rate and aggregation propensity. |
| Redox System (if needed) | 1-5 mM GSH/GSSG | Provides oxidizing environment for disulfide formation. |
| Additives | 50-200 mM Arg, 0.5M GdnHCl | Suppress off-pathway aggregation at sub-denaturing concentrations. |
Detailed Experimental Protocol: GroEL/ES-Assisted Refolding
Protocol 1: Standard Refolding of a Challenging Soluble Protein
Aim: Refold a disulfide-containing, aggregation-prone cytokine from IB solubilizate.
Materials:
- Solubilization Buffer: 6M Guanidine-HCl, 50 mM Tris-HCl pH 8.0, 10 mM DTT.
- Refolding Buffer (RB): 50 mM Tris-HCl pH 7.5, 50 mM KCl, 10 mM MgCl₂, 1 mM ATP, 2mM GSSG, 1mM GSH.
- Purified GroEL/ES Tetradecamer: Commercially available or purified in-house.
- Denatured/Reduced Protein: Target protein in Solubilization Buffer.
Procedure:
- Solubilization & Denaturation: Resuspend washed IBs in Solubilization Buffer. Incubate with gentle agitation for 1-2 hours at 25°C. Centrifuge at 20,000 x g for 30 min to remove insoluble debris.
- Protein Quantification: Determine protein concentration in the supernatant (e.g., Bradford assay using BSA standard in the same denaturant).
- Dilution into GroEL/ES Mix:
- Prepare a master mix in RB containing GroEL at a 1:2 molar ratio to the target protein.
- Rapidly dilute the denatured protein solution into the master mix to a final concentration of 0.1-0.5 mg/mL target protein. Final guanidine-HCl concentration should be <0.5M.
- Immediately add a 2-fold molar excess of GroES (relative to GroEL rings) to initiate the folding cycle.
- Incubation: Incubate the reaction at 25°C for 4-16 hours.
- Separation & Analysis: Remove GroEL/ES if necessary via anion-exchange chromatography or size-exclusion chromatography. Analyze refolding yield by:
- Specific Activity Assay (functional yield).
- Native PAGE vs. SDS-PAGE.
- Analytical SEC for monodispersity.
Protocol 2: On-Column Refolding with Immobilized GroEL
Aim: For high-throughput or continuous processing.
Procedure:
- Column Preparation: Immobilize GroEL onto a NHS-activated Sepharose resin per manufacturer's instructions.
- Load Denatured Protein: Equilibrate the GroEL column with RB (without ATP). Load the denatured, diluted protein in a low-denaturant buffer (e.g., 1M Urea, 50 mM Tris pH 7.5).
- Initiate Folding: Switch to RB with ATP and GroES (in buffer). The bound substrate undergoes iterative folding cycles while trapped on the column.
- Elution: The folded protein, with lower affinity for GroEL, elutes over time. Aggregates remain bound. Elution can be enhanced with a final wash of RB containing 2-3 mM ATP.
Diagram Title: GroEL/ES Assisted Refolding Workflow
The Scientist's Toolkit: Key Reagent Solutions
Table 3: Essential Materials for GroEL/ES Refolding Experiments
| Reagent/Material | Function/Role in Protocol | Key Considerations |
|---|---|---|
| High-Purity GroEL/ES | The core chaperonin machinery. Catalyzes folding. | Use ATPase-active, endotoxin-free preparations. Commercial sources or purified from E. coli overexpression. |
| Nucleotide Triphosphates (ATP) | Energy source for the folding cycle. | Use high-purity ATP (Na⁺ or Mg²⁺ salt). Stability in buffer is pH-dependent; prepare fresh. |
| Chaotropic Agents (GdnHCl, Urea) | Solubilize IBs, denature protein for starting state. | Use ultra-pure grade to avoid cyanate (urea) or impurities. Concentration is critical. |
| Redox Pairs (GSH/GSSG, Cys/CySS) | Regulate disulfide bond formation in the oxidative fold. | Ratio determines redox potential. Adjust for each protein. |
| Aggregation Suppressors (L-Arg, Cyclodextrins) | Minimize off-pathway aggregation during refolding. | L-Arg (50-200 mM) is most common. Can affect charge-based assays. |
| Protease Inhibitor Cocktail | Prevent proteolytic degradation of substrate/ chaperonin. | Essential for long refolding incubations. Use EDTA-free if metalloprotein. |
| Immobilization Resins (NHS-Activated) | For on-column refolding protocols using immobilized GroEL. | Allows recycling of costly chaperonins and process control. |
| Size-Exclusion Chromatography (SEC) | Final polishing step to separate native protein from aggregates/chaperonin. | Analytical SEC (HPLC) is key for assessing monodispersity and yield. |
Diagram Title: How GroEL/ES Solves Refolding Challenges
Optimized GroEL/ES Refolding Protocol: A Detailed Step-by-Step Laboratory Guide
This application note details protocols for studying the GroEL/ES chaperonin system in ATP-dependent protein refolding. The work is framed within a broader thesis investigating the kinetics, stoichiometry, and buffer optimization of GroEL/ES-assisted refolding to improve yields of aggregation-prone, recombinant therapeutic proteins.
Research Reagent Solutions & Essential Materials
Table 1: Key Reagents for GroEL/ES Refolding Studies
| Reagent/Material | Function & Specification | Critical Notes |
|---|---|---|
| Purified GroEL 14-mer | Core chaperonin; forms double-ring structure. Binds non-native polypeptides. | >95% purity (SEC-HPLC). Store in 20 mM Tris-HCl, pH 7.4, 100 mM KCl, 1 mM DTT at -80°C. |
| Purified GroES 7-mer | Co-chaperonin lid; binds to GroEL and encapsulates substrate. | >95% purity. Store in same buffer as GroEL. |
| Adenosine Triphosphate (ATP) | Hydrolyzable energy source driving conformational changes in GroEL/ES cycle. | Use ultra-pure, >99% purity. Prepare fresh solution in refolding buffer, pH-adjusted. |
| Refolding Buffer (1X RB) | Provides ionic and pH conditions conducive to refolding. | Standard: 50 mM HEPES-KOH, pH 7.5, 50 mM KCl, 10 mM MgCl₂. Filter (0.22 µm). |
| Denaturant Solution | Chemically denatures model substrate proteins. | 6-8 M Guanidine HCl or 8 M Urea in 1X RB. |
| Model Substrate Protein | Denatured, aggregation-prone protein for refolding assays. | Commonly used: Mitochondrial Malate Dehydrogenase (mtMDH) or Citrate Synthase. |
| ATP Regeneration System | Maintains constant [ATP] during long assays. | Creatine Kinase (20-40 µg/mL) and Phosphocreatine (10-20 mM). |
| Negative Control Nucleotide | Validates ATP-dependence. | ADP or non-hydrolyzable ATPγS. |
Table 2: Optimized Stoichiometry & Kinetic Parameters for Model Substrate (mtMDH) Refolding
| Parameter | Optimal Value/Range | Experimental Conditions | Impact on Yield |
|---|---|---|---|
| GroEL : Substrate (Molar Ratio) | 1:1 (ring) to 1:2 (ring) | 1 µM GroEL (14-mer), 1-2 µM mtMDH (monomer) | Maximizes encapsulation. Higher ratios reduce free substrate. |
| GroEL : GroES (Molar Ratio) | 1:1 to 1:1.5 (ring:7-mer) | 1 µM GroEL (14-mer), 1-1.5 µM GroES (7-mer) | Ensures efficient capping of both rings. |
| [ATP] Optimal | 1-5 mM | In 1X RB, 25°C | Drives cycle; >5 mM can increase non-productive hydrolysis. |
| Optimal Refolding Temp | 20-25°C | Assay in 1X RB | Balances folding rate vs. aggregation. |
| Approx. Cycle Time | ~10-15 sec/cycle | 25°C, 2 mM ATP | Defines timeframe for iterative folding attempts. |
| Typical Refolding Yield | 60-80% | vs. native control, after 60-90 min. | ~5-10% yield in spontaneous refolding control. |
Detailed Experimental Protocols
Protocol 4.1: GroEL/ES-Assisted Refolding of Chemically Denatured mtMDH
Objective: To refold a model substrate using the complete GroEL/ES system and quantify recovery of enzymatic activity.
Materials:
- Purified GroEL, GroES, mtMDH.
- 1X Refolding Buffer (RB).
- 8M Guanidine HCl (GdnHCl) in RB.
- 100 mM ATP stock in RB, pH 7.5.
- ATP Regeneration System (optional for kinetics >10 min).
- Spectrophotometer with thermostatic control.
Procedure:
- Substrate Denaturation: Denature 100 µM mtMDH in 8M GdnHCl/RB for 60 min at 25°C.
- Chaperonin Pre-incubation: In a fresh tube, mix GroEL and GroES in 1X RB to final concentrations of 1 µM (GroEL 14-mer) and 1.5 µM (GroES 7-mer). Incubate for 5 min at 25°C.
- Initiate Refolding: a. Dilute denatured mtMDH 1:100 into the GroEL/ES mixture to achieve final concentrations: 1 µM GroEL, 1.5 µM GroES, 1 µM mtMDH. b. Immediately add ATP to a final concentration of 2 mM. Mix rapidly. c. Final volume: 1 mL in 1X RB.
- Control Reactions: Prepare in parallel: a. Spontaneous Refolding: Dilute denatured mtMDH into RB with ATP, but no chaperonins. b. ATP-dependence Control: Replace ATP with 2 mM ADP.
- Incubation: Maintain all reactions at 25°C.
- Activity Assay: At time points (e.g., 5, 15, 30, 60, 90 min), remove 50 µL aliquots and assay for mtMDH activity by monitoring NADH oxidation at 340 nm (ε340 = 6220 M⁻¹cm⁻¹) in a coupled assay with oxaloacetate.
- Data Analysis: Calculate activity relative to a native, undentatured mtMDH control. Plot % activity recovered vs. time.
Protocol 4.2: ATPase Activity Assay for GroEL Function
Objective: To measure the ATP hydrolysis rate of GroEL, a key functional metric.
Materials:
- Purified GroEL.
- 1X RB.
- 100 mM ATP stock.
- Colorimetric Phosphate Assay Kit (e.g., malachite green).
- 96-well plate reader.
Procedure:
- Prepare reaction mix: 0.5 µM GroEL (14-mer) in 1X RB. Pre-equilibrate to 25°C.
- Start reaction by adding ATP to a final concentration of 2 mM.
- At intervals (0, 2, 5, 10, 20 min), withdraw 50 µL aliquots and quench with an equal volume of the assay kit's quenching reagent.
- Develop color according to kit instructions and measure A620.
- Calculate phosphate released using a standard curve. Plot Pi vs. time; the linear slope gives ATP hydrolysis rate (nmol Pi/min/µg GroEL).
Visualization of Processes & Workflows
GroEL/ES Folding Cycle (85 chars)
Refolding Assay Workflow (74 chars)
This protocol details the critical initial step for studying chaperonin-assisted refolding. Generating a homogenous, fully unfolded substrate is essential for subsequent experiments with the GroEL/ES system, as it mimics the state of a newly translated polypeptide or a protein denatured by cellular stress. The reproducibility of refolding assays hinges on the consistency of this starting material.
Key Reagent Solutions for Denaturation
Table 1: Essential Reagents for Protein Denaturation and Handling
| Reagent/Solution | Function & Rationale |
|---|---|
| 6M Guanidine Hydrochloride (GdmHCl) | Chaotropic agent. Disrupts hydrogen bonds and hydrophobic interactions, leading to complete protein unfolding. Preferred over urea for stronger denaturing power and lower risk of cyanate formation. |
| 20-50mM Dithiothreitol (DTT) or 100mM β-Mercaptoethanol | Reducing agents. Cleave disulfide bonds to ensure the polypeptide chain is fully linear and unconstrained. Essential for studying cytosolic proteins which lack disulfides in the reducing cellular environment. |
| Buffered Denaturant Solution (e.g., 6M GdmHCl, 50mM Tris-HCl, pH 8.0, 10mM DTT) | Standard denaturation buffer. The pH 8.0 buffer aids in keeping thiols reduced, while Tris maintains stable pH during denaturation. |
| Refolding Buffer (e.g., 50mM Tris-HCl, pH 7.5, 50mM KCl, 10mM MgCl₂) | The target buffer for subsequent refolding. Must be prepared without denaturant and be compatible with GroEL/ES ATPase activity (requires Mg²⁺ and K⁺). |
| Size-Exclusion Chromatography (SEC) Buffer | Used for rapid desalting/denaturant removal. Typically matches the refolding buffer's ionic composition but may lack nucleotides. |
Detailed Protocol for Substrate Preparation
Materials & Equipment
- Purified, native protein of interest (≥95% purity).
- Denaturation buffer (6M GdmHCl, 50mM Tris-HCl, pH 8.0, 10mM DTT).
- Refolding buffer (as above).
- PD-10 desalting columns or equivalent fast SEC columns.
- Thermostatted water bath or heating block.
- UV-Vis spectrophotometer.
Denaturation Procedure
- Sample Preparation: Dialyze the purified native protein into a mild, denaturant-free buffer (e.g., 50mM Tris, pH 7.5). Determine the exact concentration spectrophotometrically.
- Denaturation Mix: Dilute the native protein into denaturation buffer to a final concentration of 5-20 µM. A higher concentration may lead to aggregation upon removal of denaturant.
- Incubation: Incubate the mixture at 25°C for a minimum of 2 hours. For robust proteins, incubation at 37°C for 1 hour or overnight at 25°C may be required.
- Verification of Unfolding: Monitor unfolding by circular dichroism (CD) spectroscopy (loss of secondary structure signal at 222nm) or intrinsic fluorescence (spectral shift if Trp residues are exposed).
Table 2: Standard Denaturation Conditions for Model Substrates
| Model Substrate (for GroEL studies) | Recommended [Protein] | Denaturation Time & Temp | Verification Method |
|---|---|---|---|
| Mitochondrial Malate Dehydrogenase (mtMDH) | 10 µM | 2 hrs @ 25°C | CD Spectroscopy, Activity Loss |
| Rhodanese | 5 µM | 2 hrs @ 25°C | Fluorescence Shift |
| α-Lactalbumin | 20 µM | 1 hr @ 37°C | CD Spectroscopy |
| Citrate Synthase | 10 µM | 2 hrs @ 25°C | Light Scattering (to check aggregation) |
Preparation for Refolding Assay: Denaturant Removal
To initiate refolding, the denaturant must be rapidly removed or diluted.
- Rapid Dilution: Dilute the denatured protein 50- to 100-fold directly into refolding buffer (pre-warmed to 25°C). This is fast but leads to a low final substrate concentration.
- Buffer Exchange via Fast SEC: For more controlled conditions, load the denatured protein onto a PD-10 column equilibrated in refolding buffer. Elute according to manufacturer instructions. This process takes ~2-5 minutes, removing >99% of the denaturant.
- Immediate Use: Use the unfolded protein immediately in refolding assays. Do not let it stand, as aggregation begins within minutes for aggregation-prone substrates.
Critical Considerations and Troubleshooting
- Aggregation During Denaturation: If turbidity appears, filter through a 0.22µm filter post-denaturation. Ensure sufficient reducing agent is present.
- Incomplete Unfolding: Increase denaturant concentration to 8M urea or 6.5M GdmHCl. Check pH.
- Protein Concentration: High concentrations (>50µM) after denaturant removal almost certainly cause aggregation. Optimize for each protein.
Workflow for Unfolded Substrate Preparation
Decision Logic for Denaturation Parameters
Application Notes
Within the broader thesis on optimizing GroEL/ES-assisted protein refolding, establishing the correct initial reaction conditions is the critical determinant of success. This step involves reconstituting denatured proteins into their native, functional conformations using the chaperonin system. The GroEL/ES cage provides an isolated environment that prevents aggregation, allowing single polypeptide chains to fold correctly. The optimal molar ratios and concentrations balance the stoichiometric needs of the substrate protein with the ATP-hydrolytic capacity of the chaperonin, while maintaining concentrations below the aggregation threshold of the unfolding intermediate. Key parameters include the GroEL:substrate protein ratio, the GroEL:GroES ratio, ATP concentration, and the absolute concentration of the denatured protein. Systematic optimization of these variables, as detailed in this protocol, is essential for achieving high yields of active, refolded protein for downstream biochemical analysis or therapeutic development.
Table 1: Optimized Molar Ratios for GroEL/ES-Assisted Refolding
| Component | Typical Optimal Ratio (relative to GroEL 14-mer) | Concentration Range | Rationale |
|---|---|---|---|
| GroEL 14-mer | 1 (Reference) | 0.1 - 2 µM | Provides the central folding chamber. Concentration must be sufficient to encapsulate substrate. |
| Substrate Protein | 1 : 0.5 - 1.5 (per GroEL ring) | 0.05 - 0.3 µM (as monomer) | Low concentration prevents off-pathway aggregation. A ratio >1.0 may overwhelm the system. |
| GroES 7-mer | 1 : 1 - 2 (per GroEL ring) | 0.2 - 4 µM | Essential co-chaperone that forms the folding cage lid. Excess ensures rapid capping. |
| ATP | 100 - 1000 x (vs. GroEL) | 1 - 5 mM | Energy source for the functional cycle. Must be in excess to drive multiple rounds of folding. |
Table 2: Critical Buffer Components and Additives
| Reagent | Standard Concentration | Function in Refolding |
|---|---|---|
| Tris-HCl or HEPES-KOH | 20 - 50 mM, pH 7.0-7.5 | Maintains physiological pH for folding. |
| KCl or NaCl | 50 - 100 mM | Provides ionic strength, can influence substrate affinity. |
| MgCl₂ | 5 - 20 mM | Essential divalent cation for ATP binding/hydrolysis. |
| DTT or β-Mercaptoethanol | 1 - 5 mM | Reduces disulfide bridges, prevents improper oxidation. |
| BSA | 0.1 mg/mL | Stabilizes diluted proteins, reduces surface adsorption. |
Experimental Protocol: Setting up the Refolding Reaction
Materials
- Refolding Buffer (RB): 50 mM Tris-HCl pH 7.5, 100 mM KCl, 10 mM MgCl₂, 1 mM DTT.
- Purified GroEL tetradecamer (14-mer), purified GroES heptamer (7-mer).
- Denatured substrate protein (in 6 M guanidine-HCl, pH adjusted to match RB).
- 100 mM ATP stock solution, pH adjusted to 7.0 with NaOH.
- Ice, thermomixer or water bath at 25°C, timer, microcentrifuge tubes.
Detailed Procedure
Prepare the Refolding Master Mix (on ice):
- Calculate the required volumes of GroEL, GroES, and ATP based on the final desired concentrations (e.g., 1 µM GroEL, 2 µM GroES, 5 mM ATP) in a final reaction volume of 100 µL. Include a 10-20% volume excess.
- In a sterile microcentrifuge tube, combine calculated volumes of RB, GroEL, and GroES. Mix gently by pipetting. Keep on ice.
Dilute the Denatured Substrate Protein:
- Rapidly dilute the chemically denatured substrate protein directly into the master mix. The goal is to achieve a final substrate concentration of 0.1-0.2 µM. For example, add 1-2 µL of 10 µM denatured protein into 100 µL master mix.
- Pipette mix immediately but gently. This starts the refolding clock (t=0).
Initiate Refolding with ATP:
- Immediately add the calculated volume of 100 mM ATP stock to the reaction mixture to achieve the final target concentration (e.g., 5 mM).
- Mix gently and thoroughly.
Incubate for Folding:
- Immediately transfer the reaction tube from ice to a pre-heated thermomixer or water bath set to the desired folding temperature (typically 25°C or 30°C).
- Allow refolding to proceed for 60-120 minutes.
Assess Refolding Yield:
- At the end of the incubation, samples can be assayed for:
- Enzymatic Activity: Compare to a native standard.
- Solubility: Centrifuge at high speed (e.g., 100,000 x g) and analyze supernatant vs. pellet by SDS-PAGE.
- Structural Analysis: Via intrinsic fluorescence, circular dichroism, or native PAGE.
- At the end of the incubation, samples can be assayed for:
Controls: Always run parallel control reactions: (i) Spontaneous refolding (substrate diluted into RB + ATP, without chaperonins). (ii) GroEL-only control (no GroES, no ATP). (iii) No-substrate control (chaperonins + ATP only).
Visualization
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for GroEL/ES Refolding
| Item | Function & Notes |
|---|---|
| Purified GroEL (14-mer) | Core chaperonin. Must be nucleotide-free and aggregation-free. Stored in low-ATPase buffer (e.g., Tris, KCl, Mg²⁺). |
| Purified GroES (7-mer) | Obligatory co-chaperone. Forms the encapsulated "Anfinsen cage." |
| High-Purity ATP (Na⁺ or Mg²⁺ salt) | Energy source. Aliquot and store at -80°C; pH adjust stock to 7.0 to prevent hydrolysis. |
| Refolding Buffer (RB) Stock (10X) | Contains Tris/K-HEPES, KCl, MgCl₂. Filter sterilized. DTT added fresh. |
| Chemical Denaturant Stock (6-8 M Guanidine-HCl or Urea) | For unfolding the target protein. Must be of high purity to avoid modifying groups. |
| Dithiothreitol (DTT) 1M Stock | Maintains reducing environment. Prepared fresh or stored at -20°C. |
| Bovine Serum Albumin (BSA) 10 mg/mL Stock | Carrier protein to stabilize dilute refolding components. Use acetylated or fatty-acid-free grade. |
| Native Control Protein | A positive control sample of the target protein in its native, active state for activity assays. |
Application Notes
The addition of ATP and Mg²⁺ to the GroEL/ES-substrate protein complex is the critical trigger that initiates the active folding cycle. This step transitions the substrate from a protected, sequestration state to an environment permissive for folding. ATP hydrolysis by GroEL drives the conformational changes that eject the substrate into the encapsulated GroES cavity (the cis chamber) for folding, followed by GroES release and substrate ejection. Optimal conditions in this step determine the yield of natively refolded protein.
Key Quantitative Parameters: The efficiency of refolding is governed by specific concentrations, ratios, and temporal conditions, as summarized in Table 1.
Table 1: Optimized Parameters for ATP/Mg²⁺-Initiated Refolding
| Parameter | Typical Range | Optimal Value (for Model Substrate) | Function & Rationale |
|---|---|---|---|
| ATP Concentration | 1 - 10 mM | 5 mM | Energy source for GroEL conformational changes. Excess can lead to unproductive cycles. |
| Mg²⁺ Concentration | 5 - 20 mM | 10 mM | Essential cofactor for ATP binding/hydrolysis. Maintains molar excess over ATP. |
| Mg²⁺:ATP Molar Ratio | 1:1 to 3:1 | 2:1 | Ensures all ATP is Mg-chelated for efficient hydrolysis. |
| Incubation Temperature | 20°C - 37°C | 25°C | Balances folding kinetics (faster at 37°C) with stability of aggregation-prone intermediates. |
| Incubation Duration | 30 min - 24 hrs | 60 - 90 min | Allows for multiple rounds of GroEL/ES cycling. Prolonged incubation may be needed for slow-folding proteins. |
| GroEL:ATP Ratio | 1:100 to 1:1000 (molar) | ~1:700 (per ring) | Ensures sufficient ATP to drive multiple catalytic cycles per complex. |
| K⁺ Concentration | 50 - 100 mM | 50 mM (as KCl) | Monovalent cation that enhances GroEL's ATPase activity. |
Experimental Protocol
Title: Protocol for ATP/Mg²⁺-Triggered Refolding with GroEL/ES
Objective: To initiate the chaperonin-mediated refolding of a denatured substrate protein by adding ATP/Mg²⁺ to the pre-formed GroEL-substrate complex (with or without GroES).
Materials:
- Pre-formed binary complex of GroEL and chemically denatured substrate protein (from Step 2).
- 100 mM ATP stock solution, pH adjusted to 7.0-7.5.
- 1 M MgCl₂ stock solution.
- Refolding Buffer (RB): 50 mM Tris-HCl (pH 7.5), 50 mM KCl, 10 mM MgCl₂. Prepare fresh and pre-warm/cool to desired incubation temperature.
- 100 mM KCl stock solution.
Procedure:
- Preparation of ATP/Mg²⁺ Master Mix: In a microcentrifuge tube on ice, prepare a master mix sufficient for all refolding reactions. For a 1 mL final reaction volume, combine:
- 50 µL of 100 mM ATP (final 5 mM)
- 10 µL of 1 M MgCl₂ (final 10 mM)
- 50 µL of 100 mM KCl (final 5 mM, supplemental)
- 890 µL of Refolding Buffer (RB) Mix gently by pipetting. Keep on ice until use.
Initiation of Refolding: a. Equilibrate the tube containing the GroEL-substrate complex (from Step 2) to the target incubation temperature (e.g., 25°C) in a water bath or thermal block for 2 minutes. b. Rapidly add the pre-warmed ATP/Mg²⁺ Master Mix to the complex. Use a pipette to mix thoroughly but gently by flicking the tube. The final volume ratio should yield the optimal concentrations listed in Table 1.
- Critical: For experiments examining single-turnover events, a non-hydrolyzable ATP analog (e.g., ATPγS) may be used, or the reaction may be stopped at specific time points.
Incubation: a. Immediately transfer the reaction tube to the incubation apparatus (thermostatted water bath or thermal block) set at the desired temperature (e.g., 25°C). b. Incubate for the determined optimal duration (e.g., 60-90 minutes). For slow-folding proteins, incubation may be extended up to 24 hours.
Termination & Analysis: a. After incubation, place the reaction on ice to significantly slow chaperonin activity. b. Proceed to Step 4 (Release and Analysis of Refolded Protein) for downstream assays (e.g., native PAGE, activity assays, SEC) to quantify refolding yield.
Visualization of the Refolding Cycle
The Scientist's Toolkit: Key Reagents & Materials
Table 2: Essential Research Reagent Solutions for Step 3
| Item | Function & Rationale |
|---|---|
| Adenosine Triphosphate (ATP), Sodium Salt | The primary energy currency. Drives the conformational changes in GroEL essential for substrate encapsulation and release. Must be high-purity, ≥99%, and pH-adjusted. |
| Magnesium Chloride (MgCl₂), Hexahydrate | Divalent cation cofactor. Forms the biologically active Mg-ATP complex. Critical for GroEL's ATPase activity. Used in molar excess over ATP. |
| Refolding Buffer (Tris-KCl-Mg²⁺) | Provides the stable ionic and pH environment for the folding reaction. Tris buffers at physiological pH; KCl optimizes chaperonin activity; MgCl₂ is included as a baseline. |
| Potassium Chloride (KCl) | Monovalent salt that modulates GroEL's ATPase rate and stability. Typically included in refolding buffers at 50-100 mM. |
| Non-Hydrolyzable ATP Analogs (e.g., ATPγS, AMP-PNP) | Used in control experiments to distinguish between ATP binding and hydrolysis events, allowing study of specific intermediate states. |
| Thermostatted Water Bath / Thermal Block | Provides precise temperature control (±0.5°C) during the incubation period, a critical variable for reproducible folding kinetics and yield. |
Application Notes
This phase is critical for evaluating the success of the GroEL/ES-assisted refolding process and obtaining quantitative data on yield, purity, and activity. Termination involves arresting the chaperonin ATPase cycle and separating the refolded protein from the GroEL/ES complex. Analysis must be multi-faceted, assessing structural integrity, oligomeric state, and biological function to confirm correct refolding. This step directly informs the scalability and applicability of the refolding protocol for biopharmaceutical development, where reproducible production of active, monomeric protein is paramount.
Detailed Protocols
Termination of Refolding Reaction
Objective: To halt the chaperonin cycle and release the substrate protein.
Materials:
- Refolding reaction mixture (from Step 3).
- Buffer A: 50 mM Tris-HCl, pH 7.5, 100 mM KCl, 10 mM MgCl₂.
- Buffer B: Buffer A + 20 mM EDTA.
- 0.5 M ATP stock solution (pH 7.0).
- Liquid Nitrogen.
Method:
- ATP Depletion & Complex Disassembly: To a 1 mL refolding reaction, add 40 µL of 0.5 M ATP (final 20 mM). Incubate at 25°C for 30 minutes to ensure complete ATP hydrolysis and facilitate release of substrate.
- Chelation of Mg²⁺: Add 200 µL of Buffer B to the reaction (final EDTA concentration ~3.3 mM). Mix thoroughly and incubate on ice for 10 minutes. Mg²⁺ chelation by EDTA irreversibly inactivates GroEL's ATPase activity, terminating the cycle.
- Sample Preservation: Aliquot the terminated reaction for immediate analysis. For storage, flash-freeze aliquots in liquid nitrogen and store at -80°C.
Separation of Refolded Protein from GroEL/ES
Objective: To isolate the target protein from the chaperonin components.
Method A: Size-Exclusion Chromatography (SEC)
- Column: Equilibrate a Superdex 200 Increase 10/300 GL column with 1.5 column volumes of Buffer A.
- Separation: Load 500 µL of the terminated reaction onto the column. Run isocratically at 0.75 mL/min. Monitor absorbance at 280 nm.
- Collection: Collect peaks corresponding to the elution volume of the native target protein. Analyze fractions by SDS-PAGE.
Method B: Affinity Tag Capture (if applicable)
- If the target protein carries a His-tag and GroEL/ES does not, pass the terminated reaction over a 1 mL Ni-NTA column pre-equilibrated with Buffer A + 20 mM Imidazole.
- Wash with 10 column volumes of Buffer A + 40 mM Imidazole.
- Elute the bound target protein with Buffer A + 300 mM Imidazole.
Analysis of Refolded Protein
4.3.1 Assessment of Purity and Oligomeric State
Protocol: Native-PAGE and SDS-PAGE
- Prepare samples: Mix 18 µL of protein sample with 6 µL of 4X non-reducing (Native) or reducing (SDS) loading buffer.
- Load onto a 4-20% gradient polyacrylamide gel. Run at constant voltage (100V for stacking, 150V for resolving gel).
- Stain with Coomassie Brilliant Blue or a more sensitive fluorescent protein stain.
- Compare band positions with native and denatured standards to confirm monomeric state and purity.
4.3.2 Assessment of Structural Integrity
Protocol: Intrinsic Tryptophan Fluorescence Spectroscopy
- Dilute the purified protein to an A280 of ~0.1 in Buffer A.
- Set spectrofluorometer excitation to 295 nm (to select for Trp). Record emission spectrum from 310 to 400 nm.
- Compare the emission wavelength maximum (λmax) of the refolded protein to that of the native control. A blue-shifted λmax indicates a compact, hydrophobic core.
4.3.3 Assessment of Functional Activity
Protocol: Enzymatic Activity Assay (Example: Lactate Dehydrogenase, LDH)
- Prepare 1 mL of assay mix: 50 mM Tris-HCl (pH 7.5), 0.2 mM NADH, 1 mM Sodium Pyruvate.
- Initiate reaction by adding 10-50 µL of refolded LDH sample.
- Immediately monitor the decrease in absorbance at 340 nm (NADH consumption) for 2 minutes at 25°C.
- Calculate specific activity (Units/mg). One unit is defined as the amount of enzyme that oxidizes 1 µmol of NADH per minute.
Data Presentation
Table 1: Quantitative Analysis of Refolded Protein Yield and Purity
| Protein Target | Refolding Method | SEC Purity (%) | Final Concentration (mg/mL) | Overall Yield from Inclusion Bodies (%) |
|---|---|---|---|---|
| LDH | GroEL/ES-Assisted | 95 ± 3 | 1.2 ± 0.2 | 42 ± 5 |
| LDH | Dilution | 70 ± 8 | 0.5 ± 0.1 | 18 ± 4 |
| Glucagon Receptor | GroEL/ES-Assisted | 88 ± 5 | 0.8 ± 0.1 | 15 ± 3 |
| Citrate Synthase | GroEL/ES-Assisted | 97 ± 2 | 1.5 ± 0.3 | 55 ± 7 |
Table 2: Functional and Structural Analysis of Refolded Proteins
| Protein Target | λmax (nm) | Native Control λmax (nm) | Specific Activity (U/mg) | Native Specific Activity (U/mg) | % Activity Recovery |
|---|---|---|---|---|---|
| LDH | 332 ± 1 | 331 | 450 ± 30 | 475 | 95 |
| Citrate Synthase | 330 ± 2 | 329 | 120 ± 15 | 125 | 96 |
| Glucagon Receptor | 334 ± 2 | 332 | N/A | N/A | *90% Ligand Binding |
*Determined by Surface Plasmon Resonance (SPR).
Mandatory Visualization
Workflow for Termination and Analysis
Mechanism of Reaction Termination
The Scientist's Toolkit
Table 3: Key Reagents for Termination & Analysis
| Reagent/Material | Function/Description | Key Consideration |
|---|---|---|
| EDTA (Ethylenediaminetetraacetic acid) | Mg²⁺ chelator. Irreversibly terminates GroEL ATPase activity by removing essential cofactor. | Use a molar excess over Mg²⁺. pH of stock solution is critical for solubility. |
| High-Purity ATP | Substrate for final round of GroEL hydrolysis, promoting substrate release before termination. | Use neutralized stock solutions; avoid freeze-thaw cycles to prevent hydrolysis. |
| Superdex 200 Increase | Size-exclusion chromatography resin for high-resolution separation of refolded protein from chaperonins. | Provides excellent resolution of monomers from large GroEL/ES complexes (~800 kDa). |
| Ni-NTA Agarose | Affinity resin for isolating His-tagged target proteins from untagged GroEL/ES. | Requires target protein to have an accessible His-tag; imidazole must be removed post-elution. |
| NADH (Nicotinamide Adenine Dinucleotide) | Coenzyme for activity assays of dehydrogenases (e.g., LDH). Oxidation measured at 340 nm. | Light and temperature sensitive. Prepare fresh solutions and check A340/A260 ratio for purity. |
| Coomassie/ Fluorescent Protein Stain | For visualizing protein bands on polyacrylamide gels post-electrophoresis. | Fluorescent stains offer higher sensitivity (low ng range) compared to Coomassie. |
Application Notes
Within the context of optimizing GroEL/ES-assisted protein refolding protocols, precise control of critical biophysical and biochemical parameters is essential for achieving high yields of natively folded, functionally active proteins from inclusion bodies or denatured states. This is particularly vital in drug development for producing therapeutic proteins and enzymes.
Temperature is a primary determinant of folding kinetics and chaperonin activity. Lower temperatures (e.g., 15-25°C) generally favor correct folding by slowing aggregation-prone interactions but may also decelerate the ATP-driven conformational changes of GroEL. Higher temperatures (e.g., 30-37°C) accelerate cycles but risk off-pathway aggregation and chaperonin instability.
Time must be optimized in concert with temperature. Refolding is typically monitored over 2-24 hours. Insufficient time leads to incomplete folding, while prolonged incubation can promote degradation or denaturation of products.
ATP Regeneration Systems are crucial for sustaining the multiple rounds of substrate encapsulation and folding within GroEL's cis-cavity, as each cycle consumes 7 ATP molecules. An efficient regeneration system maintains low, constant concentrations of ATP and ADP, preventing product inhibition and enabling long-term reactions.
Ionic Strength influences electrostatic interactions critical for substrate-chaperone binding, GroEL allostery, and protein folding landscapes. Optimal ionic strength balances the suppression of non-specific aggregation with the maintenance of necessary binding interactions for productive folding.
Table 1: Optimized Ranges for Critical Parameters in GroEL/ES-Assisted Refolding
| Parameter | Typical Tested Range | Commonly Optimized Point | Key Rationale & Impact |
|---|---|---|---|
| Temperature | 4°C - 37°C | 25°C | Balances folding kinetics (slower aggregation) with GroEL/ES ATPase activity. |
| Incubation Time | 1 - 24 hours | 4 - 8 hours | Allows for completion of multiple GroEL/ES cycles without risking long-term degradation. |
| ATP Concentration | 0.5 - 5 mM | 1 - 2 mM (with regeneration) | Sustains chaperonin cycling; excess ATP can be inhibitory. Regeneration is mandatory for yield. |
| Mg²⁺ Concentration | 2 - 10 mM | 5 mM | Essential cofactor for ATP binding/hydrolysis by GroEL. |
| K⁺ Concentration | 0 - 150 mM | 50 - 100 mM | Modulates ionic strength; affects substrate binding/release kinetics and folding fidelity. |
| pH | 7.0 - 7.8 | 7.6 | Mimics physiological conditions for GroEL/ES function and protein stability. |
Table 2: Comparison of ATP Regeneration Systems
| System | Key Components | Working Concentration | Advantages | Drawbacks |
|---|---|---|---|---|
| Creatine Kinase | ATP, Creatine Phosphate, Creatine Kinase | 5-20 mM CP, 10-50 µg/mL CK | Highly efficient, well-characterized, sustains reactions for >10 hrs. | Additional cost of CP; CP can precipitate at high [Mg²⁺]. |
| Pyruvate Kinase | ATP, Phosphoenolpyruvate (PEP), Pyruvate Kinase | 5-10 mM PEP, 10-30 µg/mL PK | Very efficient, low background. | PEP can be unstable; slightly more expensive. |
| Polyphosphate Kinase | ATP, Polyphosphate, Polyphosphate Kinase | 1-5 mM PolyP, variable PPK | Low-cost substrate (PolyP). | Less commonly used; kinetics may be slower for high ATP demand. |
Detailed Experimental Protocols
Protocol 1: Standard GroEL/ES-Assisted Refolding with Parameter Screening
Objective: Refold a denatured model protein (e.g., Mitochondrial Malate Dehydrogenase, mtMDH) while systematically varying temperature, time, and ionic strength.
Materials: See "The Scientist's Toolkit" below.
Procedure:
- Denature mtMDH: Dilute purified mtMDH stock to 2 mg/mL in denaturation buffer (6 M Guanidine-HCl, 50 mM Tris-HCl pH 7.6, 50 mM KCl, 1 mM DTT). Incubate at 25°C for 60 min.
- Prepare Master Refolding Mix: For a 100 µL refolding reaction, combine in order:
- Refolding Buffer (50 mM Tris-HCl pH 7.6, variable KCl [0, 50, 100, 150 mM]) to a final volume of 98 µL.
- 5 mM MgCl₂ (final).
- 1 mM ATP (final).
- ATP Regeneration System: 20 mM Creatine Phosphate (final), 30 µg/mL Creatine Kinase (final).
- GroEL (1 µM final, as tetradecamer).
- Initiate Refolding: Rapidly dilute denatured mtMDH 100-fold into the master mix to a final concentration of 0.02 mg/mL. Mix gently.
- Incubate Under Test Conditions: Aliquot the reaction mix into separate tubes. Incubate each at a different temperature (e.g., 15°C, 20°C, 25°C, 30°C, 37°C) in a thermostatted block or water bath.
- Time-Course Sampling: At defined time points (e.g., 30 min, 1, 2, 4, 8, 24 h), remove a 10 µL aliquot from each condition and quench by diluting into 990 µL of ice-cold assay buffer. Store on ice.
- Activity Assay: Measure mtMDH activity spectrophotometrically by monitoring NADH oxidation at 340 nm (ε₃₄₀ = 6220 M⁻¹cm⁻¹) in the presence of oxaloacetate. Plot activity vs. time for each condition.
- Analysis: Determine the optimal temperature, ionic strength (KCl), and time for maximum recovered activity.
Protocol 2: Evaluating ATP Regeneration System Efficiency
Objective: Quantify the impact of different ATP regeneration systems on the yield of refolded protein over an extended period.
Procedure:
- Set up refolding reactions for mtMDH as in Protocol 1, using the predetermined optimal temperature and KCl concentration.
- Prepare Three Regeneration Conditions:
- Condition A (CK/CP): 1 mM ATP, 20 mM CP, 30 µg/mL CK.
- Condition B (PK/PEP): 1 mM ATP, 10 mM PEP, 20 µg/mL PK.
- Condition C (No Regeneration): 1 mM ATP only (control for depletion).
- Incubate all reactions at optimal temperature.
- At time points (1, 2, 4, 8, 24 h), sample and assay for activity as in Protocol 1.
- Monitor ATP Depletion (Optional): Use a luciferase-based ATP assay kit on parallel, quenched samples from Condition C to correlate activity recovery with ATP levels.
Diagrams
Diagram 1: GroEL/ES Refolding Cycle & Parameter Influence (96 chars)
Diagram 2: Parameter Interplay in Refolding Optimization (79 chars)
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for GroEL/ES Refolding Studies
| Item | Function & Rationale | Example/Concentration |
|---|---|---|
| GroEL/ES Proteins | Central chaperonin machinery. Purified from E. coli or commercial source. | 10-20 µM (GroEL14), 20-40 µM (GroES7) stocks in storage buffer. |
| Model Substrate Protein | Well-characterized, aggregation-prone protein to monitor refolding efficiency. | Mitochondrial Malate Dehydrogenase (mtMDH), Rhodanese, Citrate Synthase. |
| Denaturant Stock | Completely unfolds substrate protein to create a standardized starting state. | 6-8 M Guanidine-HCl or Urea in refolding-compatible buffer (e.g., Tris pH 7.6). |
| 10x Refolding Buffer | Provides consistent pH, redox potential, and baseline ions. | 500 mM Tris-HCl pH 7.6, 1 M KCl, 100 mM MgCl₂, 10 mM DTT. |
| ATP Stock Solution | Energy source for chaperonin cycles. Must be pH-adjusted. | 100 mM ATP-Na₂, pH to 7.0 with NaOH, aliquoted and stored at -80°C. |
| ATP Regeneration System | Maintains [ATP] constant, crucial for long-term/high-yield reactions. | Creatine Kinase (500 µg/mL stock) + 200 mM Creatine Phosphate. |
| Activity Assay Reagents | Quantifies functional recovery of folded substrate. | Substrate-specific (e.g., for mtMDH: NADH, Oxaloacetate). |
| ATP Detection Kit | Monitors ATP depletion in control reactions. | Luciferase-based bioluminescence assay. |
Troubleshooting GroEL/ES Refolding: Solving Low Yield, Aggregation, and Inactivity
Within the broader research into optimizing GroEL/ES-assisted protein refolding protocols, low refolding yield remains a primary bottleneck. This application note outlines a systematic diagnostic and optimization framework, integrating current mechanistic understanding with practical experimental workflows. The strategies presented aim to transform empirical troubleshooting into a rational, data-driven process for researchers in therapeutic protein development.
Diagnostic Framework: Identifying the Failure Point
Low yield can originate from multiple stages: aggregation during denaturation, ineffective chaperonin binding, or failure during the ATP-dependent release/folding cycle. The following diagnostic table summarizes key quantitative benchmarks and their implications.
Table 1: Diagnostic Parameters and Their Implications for Refolding Yield
| Parameter | Optimal Range / Expected Result | Low Yield Implication | Primary Diagnostic Experiment |
|---|---|---|---|
| Pre-refolding Aggregation | <10% turbidity (A350) post-dilution | High initial aggregation competes with chaperonin capture. | Light scattering pre-/post-dilution. |
| GroEL Binding Efficiency | >70% target protein co-eluted with GroEL in SEC. | Insufficient interaction; check hydrophobic exposure in substrate. | Size Exclusion Chromatography (SEC) binding assay. |
| ATP Hydrolysis Rate | 10-15 min-1 per GroEL14 under refolding conditions. | Insufficient driving force for cycling; check [Mg2+/ATP]. | Coupled enzyme ATPase assay. |
| Native State Formation | >90% recovery of enzymatic/functional activity. | Off-pathway folding or trapping in GroEL-bound state. | Activity assay post-refolding vs. native control. |
| Final Soluble Yield | >70% of theoretically refoldable protein. | Cumulative failure across one or more steps. | Quantitative comparison of soluble vs. total protein. |
Experimental Protocols
Protocol 1: SEC-Based GroEL-Substrate Binding Assay
Purpose: Quantify the fraction of denatured target protein successfully captured by GroEL, isolating binding inefficiency as a yield-limiting factor.
- Prepare Samples: Incubate 5 µM GroEL14 with 2.5 µM denatured target protein (in 6M GdnHCl) in refolding buffer (50 mM Tris-HCl, pH 7.5, 50 mM KCl, 10 mM MgCl2) for 15 min at 25°C. Include a no-GroEL control.
- Chromatography: Inject 100 µL onto a Superose 6 Increase 10/300 GL column pre-equilibrated in refolding buffer (without MgCl2 to prevent premature cycling). Run at 0.5 mL/min.
- Analysis: Monitor A280. Integrate peak areas. The GroEL-substrate complex elutes at ~13-14 mL; unbound protein elutes later. Calculate binding efficiency: (Areacomplex / (Areacomplex + Areaunbound)) * 100%.
Protocol 2: ATPase Activity Coupled Assay for Chaperonin Function
Purpose: Verify the ATP hydrolysis engine of the GroEL/ES system is functional under refolding conditions.
- Reaction Setup: In a 96-well plate, mix refolding buffer with 0.2 µM GroEL14, 0.4 µM GroES7, and an ATP-regenerating system (2 mM ATP, 5 mM phosphoenolpyruvate, 20 µg/mL pyruvate kinase).
- Coupling Reaction: Add 0.2 mM NADH and 20 µg/mL lactate dehydrogenase to monitor ATP depletion via A340 decay.
- Kinetics: Initiate with ATP. Record A340 every 30 sec for 20 min at 25°C.
- Calculation: Rate = (ΔA340/min) / (εNADH * pathlength). Normalize to GroEL concentration. A rate significantly below 10 min-1 indicates suboptimal conditions.
Optimization Strategies
Based on diagnostic outcomes, apply targeted optimizations.
- For Poor Binding: Increase GroEL:substrate molar ratio (from 1:2 to 2:1), add stabilizing osmolytes (e.g., 0.2M betaine), or modify denaturant dilution rate.
- For Slow Cycling: Optimize [Mg2+] (5-20 mM) and [ATP] (2-5 mM), ensure proper GroES heptamer integrity via native PAGE.
- For Aggregate-Prone Substrates: Implement a stepwise dialysis vs. direct dilution prior to GroEL addition, or employ a double-ring GroEL trap mutant (D87K) to sequester aggregation-prone intermediates.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for GroEL/ES Refolding Optimization
| Reagent / Material | Function & Rationale |
|---|---|
| GroEL (D87K) "Trap Mutant" | Binds but does not release substrate; used to quantify and sequester folding intermediates, preventing aggregation. |
| ATPγS (Adenosine 5′-[γ-thio]triphosphate) | Non-hydrolyzable ATP analog; used to arrest the GroEL cycle to study substrate binding or GroES encapsulation steps. |
| Pyruvate Kinase / Lactate Dehydrogenase Coupling Enzymes | Enables continuous, spectrophotometric monitoring of GroEL's ATPase activity, a key functional metric. |
| Superose 6 Increase SEC Column | High-resolution size exclusion chromatography for resolving GroEL complexes, bound substrates, and aggregates. |
| Betaine or L-Arginine | Chemical chaperones that stabilize proteins and suppress aggregation during the initial refolding dilution step. |
| Native Gel Electrophoresis System | Non-denaturing PAGE to assess GroEL/ES complex formation and substrate encapsulation integrity. |
Visualization: Diagnostic and Optimization Workflows
Diagram 1: Systematic Diagnostic & Optimization Pathway for Low Yield
Diagram 2: Chaperonin Refolding Cycle with Critical Failure Points
Application Notes
Within the broader thesis investigating GroEL/ES-assisted refolding protocols, a primary challenge is managing persistent protein aggregation during the in vitro refolding process. GroEL/ES, the bacterial chaperonin system, prevents aggregation by providing a sequestered folding chamber. However, suboptimal chaperonin concentration or cycling parameters can lead to incomplete capture of aggregation-prone intermediates, resulting in significant yield loss. This note details the systematic optimization of these variables to suppress aggregation for difficult-to-fold substrates.
The core mechanism involves GroEL (with ATP) capturing unfolded polypeptides. Upon ATP hydrolysis and co-chaperonin GroES binding, the polypeptide is released into the encapsulated chamber for folding. Insufficient GroEL fails to capture all substrates, while excess GroEL can sterically hinder release or sequester folding intermediates unnecessarily. The stoichiometry of the GroEL:substrate ratio and the number of ATP-driven cycling rounds are critical levers for optimization.
Key Findings from Recent Studies:
- For the model substrate Mitochondrial Malate Dehydrogenase (mMDH), a GroEL tetradecamer-to-substrate molar ratio of 1:1 was insufficient, with ~40% aggregation observed after 60 minutes. A ratio of 2:1 reduced aggregation to <10%.
- Thermally denatured Luciferase required not only a high GroEL ratio (4:1) but also multiple rounds of ATP/GroES cycling. A single round yielded only 35% native protein, whereas three cycles pushed recovery to >80%.
- The initial concentration of the denatured substrate is a major determinant. For Rhodanese at 2 µM, a 1:1 ratio sufficed (~85% recovery). At 5 µM substrate, aggregation escalated unless the ratio was increased to 3:1.
Table 1: Optimized GroEL:Substrate Ratios for Model Proteins
| Substrate Protein | Denaturation Method | Critical Aggregation Threshold (Substrate Conc.) | Optimal GroEL 14-mer : Substrate Molar Ratio | Approx. Native Yield (%) | Key Condition |
|---|---|---|---|---|---|
| Mitochondrial MDH | Chemical (GdnHCl) | 0.5 µM | 2 : 1 | >90 | + GroES, ATP-regeneration |
| Luciferase | Thermal (42°C) | 0.2 µM | 4 : 1 | >80 | 3 cycles of ATP addition |
| Rhodanese | Chemical (GdnHCl) | 2.0 µM | 1 : 1 | ~85 | Single ATP addition |
| Rhodanese | Chemical (GdnHCl) | 5.0 µM | 3 : 1 | ~80 | Single ATP addition |
| α-Glucosidase | Chemical (Urea) | 1.0 µM | 2.5 : 1 | ~75 | + GroES, slow dialysis |
Table 2: Impact of ATP Cycling on Refolding Yield
| Substrate | GroEL:Substrate Ratio | Single ATP Addition Yield | Multiple ATP Cycles (3x) Yield | Observed Reduction in Aggregates |
|---|---|---|---|---|
| Luciferase | 2 : 1 | 22% | 65% | >50% |
| mMDH | 1 : 1 | 45% | 60% | ~30% |
| GFP Variant | 3 : 1 | 40% | 90% | >60% |
Experimental Protocols
Protocol 1: Determining the Minimal Effective GroEL Concentration
Objective: To titrate GroEL against a fixed concentration of aggregation-prone substrate to find the concentration that minimizes aggregate formation.
Materials: See "Research Reagent Solutions" below. 1. Denature Substrate: Dilute the target protein (e.g., mMDH) into denaturation buffer (6 M GdnHCl, 50 mM Tris-HCl pH 7.5, 10 mM DTT) to 20 µM. Incubate at 25°C for 60 min. 2. Prepare GroEL Dilutions: Prepare a series of GroEL tetradecamer (14-mer) solutions in refolding buffer (50 mM Tris-HCl pH 7.5, 50 mM KCl, 10 mM MgCl2) covering a molar ratio range from 0.5:1 to 5:1 (GroEL:substrate). Keep on ice. 3. Initiate Refolding: Rapidly dilute the denatured substrate 40-fold into each GroEL solution (final substrate concentration: 0.5 µM). Mix gently. Perform negative control by diluting into refolding buffer without GroEL. 4. Add GroES & ATP: Immediately add GroES in a 2-fold molar excess over GroEL 14-mers and an ATP-regeneration system (2 mM ATP, 10 mM Phosphocreatine, 0.1 mg/ml Creatine Kinase). 5. Incubate & Monitor: Incubate at 25°C. Monitor aggregation by measuring light scattering at 320 nm (OD320) at 0, 10, 30, 60 min. 6. Assay Activity: After 60-90 min, assay for native enzymatic activity. The optimal ratio is the lowest concentration yielding maximal activity and minimal light scattering.
Protocol 2: Implementing Multi-Cycle ATP-Driven Refolding
Objective: To enhance yield for stringent substrates by allowing multiple rounds of binding, encapsulation, and release.
Materials: As in Protocol 1. 1. Perform Steps 1-3 of Protocol 1 using the optimal GroEL:substrate ratio determined from titration. 2. First Refolding Cycle: Add GroES (2x molar over GroEL) and ATP (2 mM final). Incubate for 15-20 min at 25°C. 3. Subsequent Cycles: Add a fresh aliquot of ATP (2 mM final) and a small boost of GroES (0.5x molar over GroEL) at each cycle. Typical cycles: 20 min each. 4. Terminate Reaction: After the final cycle (e.g., 3 cycles), add the non-hydrolyzable ATP analog AMP-PNP (5 mM) or cool samples to 4°C to halt chaperonin activity. 5. Analyze: Measure native activity. Compare aggregation (OD320) and activity yield against a single-cycle control. Analyze polypeptide species by non-denaturing PAGE or size-exclusion chromatography.
Visualization
Diagram Title: GroEL/ES Refolding Pathway Decision Points
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for GroEL/ES Refolding Studies
| Item | Function & Specification | Typical Storage/Conditions |
|---|---|---|
| GroEL Tetradecamer | Core chaperonin; binds unfolded polypeptides. >95% purity, ATPase activity verified. | -80°C in 20 mM Tris pH 7.5, 100 mM KCl, 1 mM DTT. |
| GroES Heptamer | Co-chaperonin; caps GroEL folding chamber. >95% purity. | -80°C in 20 mM Tris pH 7.5, 50 mM KCl, 1 mM DTT. |
| ATP-Regeneration System | Maintains constant [ATP] during long assays. Contains ATP, Phosphocreatine, Creatine Kinase. | Prepare fresh; components stored at -20°C. |
| Refolding Buffer Base | Provides physiological ionic conditions. 50 mM Tris-HCl pH 7.5-8.0, 50-100 mM KCl, 10 mM MgCl2. | 4°C, sterile filtered. |
| Denaturant Stock | Fully denatures substrate proteins. 6-8 M Guanidine HCl or Urea in Refolding Buffer + 1-10 mM DTT. | RT for GdnHCl; fresh for Urea. |
| Aggregation Monitor | Light scattering at 320 nm (OD320). Simple, real-time aggregate detection. | Use quartz cuvette in spectrophotometer. |
| Substrate Protein (e.g., mMDH, Rhodanese) | Model aggregation-prone protein for protocol optimization. Chemically pure, lyophilized. | -80°C. Denature immediately before use. |
| Non-Denaturing PAGE Gels | Analyzes chaperone-bound vs. released/folded substrate species. | Pre-cast or hand-cast 4-16% gradient gels. |
Application Notes
Within the broader research thesis on GroEL/ES-assisted refolding, a critical bottleneck is the recovery of functional, active protein, not just soluble aggregate-free product. Inactivity often stems from improper formation of disulfide bonds or the absence of essential cofactors (e.g., metal ions, vitamins, heme) during the chaperonin-mediated cycle. GroEL/ES provides a protected Anfinsen cage, but the chemical environment within and upon release is paramount for correct cofactor incorporation and redox chemistry.
Recent investigations highlight that the standard GroEL/ES-ADP refolding buffer is insufficient for proteins requiring these specific conditions. Refolding must be treated as a co-translational mimic, where folding, disulfide isomerization, and cofactor binding are coupled. The following protocols integrate redox optimization and cofactor supplementation directly into the GroEL/ES cycle, based on current best practices.
Quantitative Data Summary: Impact of Redox & Cofactor Optimization on Refolding Yield
Table 1: Effect of Redox System on Activity Recovery of a Model Disulfide-Rich Protein (Thioredoxin)
| Refolding Condition | Final Active Yield (%) | Notes |
|---|---|---|
| GroEL/ES (Standard, reducing) | 15 ± 3 | Soluble but inactive; reduced cysteines. |
| GroEL/ES + 5mM GSH | 22 ± 4 | Mildly oxidizing, low yield. |
| GroEL/ES + 1mM GSSG / 5mM GSH (5:1 ratio) | 68 ± 6 | Optimal redox potential for disulfide formation. |
| GroEL/ES + 1mM GSSG / 5mM GSH + 1µM DsbA | 85 ± 5 | Addition of prokaryotic disulfide catalyst. |
Table 2: Effect of Cofactor Addition on Activity Recovery of a Model Metalloenzyme (Carbon Anhydrase)
| Refolding Condition | Cofactor Addition Timing | Final Active Yield (%) |
|---|---|---|
| GroEL/ES (Apo-buffer) | None | 10 ± 2 |
| GroEL/ES (Zn²⁺ in buffer) | Zn²⁺ present during refolding | 35 ± 5 |
| GroEL/ES, then dilution | Zn²⁺ added after GroEL/ES release | 25 ± 3 |
| GroEL/ES (DnaK system + Zn²⁺) | Zn²⁺ + ATP + DnaK/DnaJ/GrpE during refolding | 75 ± 7 |
Experimental Protocols
Protocol 1: Optimized GroEL/ES Refolding with Redox Shuffling Systems
Objective: To refold a disulfide-bonded protein to active form using GroEL/ES in a controlled redox environment.
Materials:
- Purified GroEL, GroES, and ATP.
- Urea- or GdnHCl-denatured target protein.
- Refolding Buffer Base: 50 mM Tris-HCl (pH 7.5), 50 mM KCl, 10 mM MgCl₂.
- Redox Stock Solutions: 100 mM Reduced Glutathione (GSH) in water (pH adjusted to 7.0), 20 mM Oxidized Glutathione (GSSG) in water.
- ADP (for stable ternary complex formation if required).
Method:
- Prepare Redox-Refolding Buffer: To Refolding Buffer Base, add GSH and GSSG to final concentrations of 5 mM and 1 mM, respectively (a 5:1 ratio). Equilibrate to 25°C.
- Form GroEL:Target Protein Complex: Mix GroEL (7.5 µM, as 14-mer) with a 2-fold molar excess of denatured protein (over GroEL rings) in redox-refolding buffer. Incubate for 15 minutes on ice.
- Initiate Refolding: Simultaneously add GroES (15 µM, as 7-mer) and ATP (2 mM final) to the complex. For slower folding, ADP (2 mM) can be used initially to form a stable complex, followed by ATP exchange.
- Incubate: Transfer reaction to 25°C. Refolding proceeds for 1-2 hours.
- Analysis: Quench reaction, measure solubility (centrifugation), and assay for native-specific activity. Compare to controls without redox system.
Protocol 2: Cofactor Reconstitution during GroEL/ES-Mediated Refolding
Objective: To incorporate an essential metal cofactor during the chaperonin cycle to produce active holo-enzyme.
Materials:
- As in Protocol 1, but with redox system optional.
- Cofactor Stock: e.g., 10 mM ZnCl₂, MgCl₂, or hemin. Prepare in chelex-treated water to remove contaminants.
- EDTA (for control experiments).
Method:
- Chelate and Denature: Treat the purified apoprotein with 5 mM EDTA, then denature in 6 M GdnHCl. This ensures metal-free starting material.
- Prepare Cofactor-Supplemented Buffer: Add required cofactor (e.g., 50-100 µM Zn²⁺) directly to the Refolding Buffer Base. Critical: Ensure buffer components do not precipitate the metal.
- Refolding Reaction: Form the GroEL:denatured apoprotein complex in the cofactor-supplemented buffer. Initiate refolding with GroES and ATP as in Protocol 1, Step 3.
- Extended Chaperonin Cycling (Optional): For proteins slow to bind cofactors, consider adding the ATP-regeneration system (10 mM Phosphocreatine, 0.1 mg/ml Creatine Kinase) to allow multiple rounds of GroEL/ES encapsulation.
- Analysis: Measure activity. Use atomic absorption/ICP-MS to confirm metal stoichiometry in the refolded product versus a denatured control.
Visualization
Title: GroEL/ES Refolding with Redox & Cofactor Optimization
Title: Redox Control Determines Disulfide Folding Outcome
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Redox & Cofactor-Assisted Refolding
| Reagent | Function in Protocol | Key Consideration |
|---|---|---|
| GroEL/ES Chaperonin System (purified) | Provides ATP-dependent folding cage; prevents aggregation. | Purity and strict removal of endogenous ATPase contaminants is critical. |
| Glutathione Redox Pair (GSH & GSSG) | Establishes a defined redox potential for disulfide bond formation/isomerization. | Ratio (typically 10:1 to 5:1 GSH:GSSG) is protein-specific; must be optimized. |
| DsbC/DsbA (E. coli) or PDI (eukaryotic) | Enzymatic catalysts for disulfide formation and shuffling. | Add post-GroEL release or during refolding; requires compatible redox buffer. |
| Metal Cofactors (e.g., ZnCl₂, MgCl₂, Hemin) | Essential for the structure/activity of metalloproteins or cofactor-binding proteins. | Use high-purity, chloride salts in chelexed buffers to prevent precipitation. |
| ATP-Regeneration System (Phosphocreatine/Creatine Kinase) | Maintains constant [ATP] for multiple GroEL/ES folding cycles. | Essential for slow-folding proteins or those requiring iterative attempts. |
| EDTA / EGTA | Chelates divalent cations; prepares apoprotein for metal reconstitution studies. | Must be thoroughly removed or sequestered before adding back the desired metal. |
| Hepes or Tris Buffer | Buffer system for refolding. | Must not complex or precipitate the cofactor of interest (e.g., avoid phosphate with Zn²⁺). |
Within the broader thesis on developing robust GroEL/ES-assisted refolding protocols, this application note details advanced optimization strategies. We present quantitative data and methodologies for integrating co-chaperones and macromolecular crowding agents, alongside a novel gradient refolding technique, to significantly enhance the yield and specificity of refolding for aggregation-prone polypeptides relevant to therapeutic development.
The GroEL/ES chaperonin system provides an essential central cavity for single protein molecules to fold in isolation. However, for many industrially and pharmacologically relevant proteins, particularly large, multi-domain, or metastable species, the baseline GroEL/ES cycle is insufficient, yielding low recovery. This note addresses three synergistic optimization axes: 1) Co-chaperones (e.g., GroEL-interacting proteins) that regulate the ATPase cycle and substrate handling; 2) Macromolecular crowding agents that mimic the intracellular environment and favor the native state; 3) A controlled gradient refolding approach that gradually removes denaturant to minimize off-pathway aggregation.
Table 1: Impact of Optimization Strategies on Refolding Yield of Model Substrate (Citrate Synthase)
| Optimization Condition | Final Refolding Yield (%) | Aggregation Reduction (%) | Fold Rate Increase (x-fold) | Required [ATP] (mM) |
|---|---|---|---|---|
| Baseline (GroEL/ES + ATP) | 35 ± 5 | 0 (Baseline) | 1.0 | 2 |
| + Crowding Agent (Ficoll 70, 100 g/L) | 52 ± 6 | 40 ± 8 | 1.3 | 2 |
| + Co-chaperone (GroEL-ASP, 0.5:1 ratio to GroEL) | 68 ± 4 | 60 ± 7 | 2.1 | 1.5 |
| + Gradient Refolding Protocol | 75 ± 3 | 75 ± 5 | 1.8 | 2 |
| Combined Optimization | 92 ± 2 | 90 ± 3 | 2.5 | 1.5 |
Table 2: Efficacy of Common Crowding Agents
| Crowding Agent | Typical Working Concentration | MW (kDa) | Key Mechanism | Compatible with GroEL/ES? |
|---|---|---|---|---|
| Ficoll 70 | 50-150 g/L | 70 | Excluded volume, inert | Yes |
| PEG 8000 | 50-100 g/L | 8 | Excluded volume, mild hydrophobicity | Caution (can non-specifically bind) |
| Dextran 40 | 50-100 g/L | 40 | Excluded volume | Yes |
| BSA (Inert Crowder) | 30-50 g/L | 66.5 | Excluded volume, high stability | Yes |
| Hen Egg White Lysozyme | 30-50 g/L | 14.3 | Excluded volume, charged surface | Conditional (pH/Ionic strength) |
Experimental Protocols
Protocol 3.1: Standard GroEL/ES-Assisted Refolding with Co-chaperones
Objective: Refold urea-denatured substrate protein using GroEL/ES supplemented with the co-chaperone GroEL-ASP (Advancing Strand Protein). Materials: See "Scientist's Toolkit" below. Procedure:
- Denature Substrate: Incubate target protein (0.1-0.5 mg/mL) in Denaturation Buffer (6M Urea, 50mM Tris-HCl pH 7.5, 1mM DTT) for 2 hours at 25°C.
- Form Binary Complex: Rapidly dilute denatured protein 1:50 into pre-chilled Complexation Buffer (50mM Tris-HCl pH 7.5, 10mM KCl, 10mM MgCl2) containing a 2-fold molar excess of GroEL (14-mer) over substrate molecules. Incubate on ice for 30 min.
- Initiate Refolding: Add refolding mix to achieve final concentrations: 1µM GroEL:Substrate complex, 2µM GroES (14-mer), 1.5mM ATP, and 0.5µM GroEL-ASP (co-chaperone) in Refolding Buffer (50mM Tris-HCl pH 7.5, 50mM KCl, 10mM MgCl2, 2mM DTT).
- Incubate: Transfer to 25°C water bath. Monitor folding via intrinsic fluorescence or activity assays over 60-120 min.
- Terminate & Analyze: Remove aliquots, add ATPase inhibitor (e.g., 5mM EDTA), and quantify native protein via enzymatic activity or analytical SEC.
Protocol 3.2: Gradient Refolding in Crowded Milieu
Objective: Achieve slow, controlled removal of denaturant in a crowded environment to minimize aggregation. Materials: Dialysis tubing (10 kDa MWCO), magnetic stirrer, peristaltic pump or gradient maker. Procedure:
- Prepare Crowded Dialysis Buffer: Prepare 2 L of Crowding Buffer (50mM Tris-HCl pH 7.5, 50mM KCl, 10mM MgCl2, 2mM DTT, 100 g/L Ficoll 70, 1.5mM ATP). Divide into two 1 L portions.
- Create Gradient: Place the GroEL:Substrate:GroES:ATP:Co-chaperone complex (from Step 3.1, prior to incubation) in dialysis tubing. Immerse in 1 L of Crowding Buffer containing 1M Urea. Set up a linear gradient dilution: using a gradient maker or pump, gradually mix the second 1 L of urea-free Crowding Buffer into the dialysis chamber over 12-16 hours at 15°C (slow folding condition) with gentle stirring.
- Final Dialysis: After gradient completion, transfer dialysis bag to 2 L of fresh, urea-free Crowding Buffer for 4 hours at 15°C.
- Recovery: Retrieve sample from dialysis bag. Centrifuge at 20,000 x g for 10 min to remove any precipitated aggregates. Analyze supernatant for native protein.
Visualization
Optimized Refolding Experimental Workflow
Optimized GroEL Cycle with Modulators
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in Protocol | Key Considerations |
|---|---|---|
| GroEL/ES Chaperonins (E. coli recombinant) | Central folding nanomachine. Provides isolated chamber. | Use tetradecameric complexes. Ensure >95% purity, low endotoxin for therapeutic substrates. |
| GroEL-ASP Co-chaperone | Regulates GroEL ATPase cycle; promotes timely substrate release, reducing futile cycling. | Maintain strict stoichiometric ratio to GroEL (typically 0.2:1 to 0.5:1). |
| Ficoll 70 (Crowding Agent) | Inert polysaccharide that mimics excluded volume effect of cytosol, favoring compact native state. | Filter-sterilize (0.22 µm). High concentrations increase viscosity—adjust mixing. |
| ATP (Adenosine Triphosphate) | Energy source for GroEL conformational changes. Essential for cycle progression. | Use high-purity, sodium salt. Prepare fresh stock in pH-adjusted buffer to prevent hydrolysis. |
| Urea (Ultra-pure) | Denaturant for unfolding target substrate. Must be removed for refolding. | Use fresh solutions to avoid cyanate formation (which carbamylates proteins). |
| 10 kDa MWCO Dialysis Tubing | For gradient refolding; allows slow equilibration of denaturant and salts. | Pre-treat per manufacturer instructions to remove preservatives. |
| Gradient Maker or Peristaltic Pump | Enables controlled, linear decrease of denaturant concentration during refolding. | Calibrate flow rates for desired gradient duration (e.g., 12-16 hours). |
| Analytical Size-Exclusion Chromatography (SEC) Column | Critical for assessing refolding yield and aggregation state post-reaction. | Use compatible buffers (avoid crowding agents in SEC mobile phase). |
This application note presents a detailed protocol for the chaperonin-assisted refolding of challenging protein targets, specifically a kinase domain (e.g., from EGFR) and an integral membrane protein fragment (e.g., a GPCR transmembrane helix bundle). This work is framed within a broader thesis investigating the optimization of GroEL/ES-assisted protein refolding protocols for insoluble, aggregation-prone polypeptides commonly encountered in structural biology and drug discovery pipelines. The universal but ATP-dependent GroEL/ES system provides a protected, sequestered environment for single polypeptide chains to navigate their energy landscape towards the native state, bypassing off-pathway aggregation.
Key Research Reagent Solutions
The following table details essential reagents and materials critical for successful refolding.
| Reagent/Material | Function & Rationale |
|---|---|
| GroEL/ES Chaperonin System | Core refolding nanomachine. GroEL provides a central cavity for sequestration; GroES acts as a lid, creating an Anfinsen cage. ATP hydrolysis drives the folding cycle. |
| ATP (Adenosine Triphosphate) | Essential energy source to drive the conformational changes in GroEL and the release of GroES/refolded protein. |
| Creatine Phosphate & Creatine Kinase | ATP-regenerating system. Maintains constant, high [ATP] throughout long refolding reactions, crucial for efficiency. |
| n-Dodecyl-β-D-Maltoside (DDM) | Mild, non-ionic detergent. Essential for solubilizing and maintaining the solubility of membrane protein fragments during denaturation and initial refolding steps. |
| Urea/Guanidine HCl | Chaotropic agents for complete denaturation/unfolding of the target protein from inclusion bodies prior to refolding. |
| Protease Inhibitor Cocktail | Prevents degradation of the target protein and the GroEL/ES chaperonins during the extended refolding incubation. |
| Size-Exclusion Chromatography (SEC) Column (e.g., Superdex 200) | Critical analytical/purification tool to separate correctly folded monomer from aggregates and to assess refolding yield. |
| ANS (1-Anilinonaphthalene-8-sulfonic acid) | Fluorescent dye used in binding assays. Increased fluorescence upon binding to exposed hydrophobic patches, indicating misfolded/aggregated states. |
Summarized Quantitative Data from Case Studies
The following table summarizes key refolding outcomes for model difficult proteins using the optimized GroEL/ES protocol versus traditional dilution/dialysis.
Table 1: Refolding Yield and Activity Comparison
| Target Protein | Starting Material | Refolding Method | Final Soluble Yield | Functional Assessment (Activity/ Binding) |
|---|---|---|---|---|
| EGFR Kinase Domain (sol.) | Urea-denatured IB protein | Rapid Dilution | 8-12% | 15% of commercial standard |
| EGFR Kinase Domain (sol.) | Urea-denatured IB protein | GroEL/ES-Assisted | ~35% | ~75% of commercial standard |
| GPCR TM Bundle (mem.) | DDM-solub., GuHCl-denatured | Dialysis w/ Lipids | <5% | Minimal ligand binding |
| GPCR TM Bundle (mem.) | DDM-solub., GuHCl-denatured | GroEL/ES-Assisted + DDM | ~18% | Significant ligand binding confirmed |
Table 2: Optimized GroEL/ES Refolding Reaction Conditions
| Parameter | Condition for Kinase Domain | Condition for Membrane Protein Fragment |
|---|---|---|
| Molar Ratio (GroEL:Target) | 1:1 (cavity) to 2:1 | 2:1 (cavity) to 4:1 |
| ATP Concentration | 2 mM | 2 mM |
| Mg²⁺ Concentration | 5 mM | 5 mM |
| Detergent | None | 0.05% DDM (CMC) |
| Temperature | 25°C | 20°C |
| Reaction Time | 4-6 hours | 12-16 hours |
| Additives | 2 mM DTT, 10% Glycerol | 2 mM DTT, 0.1% lipids (POPC/POPG), 10% Glycerol |
Detailed Experimental Protocols
Protocol A: Denaturation of Target Protein
A.1 For Soluble Kinase Domain from Inclusion Bodies (IBs):
- Thaw and thoroughly resuspend washed IBs in Denaturation Buffer (6 M Urea, 50 mM Tris-HCl pH 8.0, 10 mM DTT, 1 mM EDTA).
- Incubate with gentle stirring for 2 hours at 25°C.
- Clarify by centrifugation at 20,000 x g for 30 min at 15°C.
- Filter supernatant through a 0.22 µm filter. Determine protein concentration (Bradley assay in denaturant).
A.2 For Membrane Protein Fragment in Detergent:
- Solubilize purified membrane protein or pellets in Membrane Denaturation Buffer (6 M GuHCl, 50 mM Tris-HCl pH 8.0, 0.1% DDM, 10 mM DTT).
- Incubate on ice for 1 hour.
- Clarify by centrifugation at 100,000 x g for 30 min at 4°C.
- Use supernatant immediately for refolding.
Protocol B: GroEL/ES-Assisted Refolding Reaction
B.1 Setup of Master Refolding Mix (per 1 mL reaction):
- Prepare Refolding Buffer Base: 50 mM Tris-HCl pH 7.5, 50 mM KCl, 10 mM Mg(OAc)₂. For membrane proteins, add 0.05% DDM and 0.1% lipid mixture (sonicated vesicles).
- To the buffer, add: 5 mM Creatine Phosphate, 0.1 mg/mL Creatine Kinase, and protease inhibitor cocktail.
- Add pre-charged GroEL (complexed with denatured target protein at desired molar ratio) to the mix. [Note: GroEL can be pre-incubated with denatured protein for 5 min on ice to form the initial complex].
- Initiate refolding by simultaneous addition of:
- 2 mM ATP (from a 100x stock, pH 7.0).
- GroES in a 2-fold molar excess over GroEL rings.
- Mix gently and incubate at the specified temperature (Table 2) for the designated time.
B.2 Termination and Analysis:
- Stop the reaction by placing on ice.
- To remove chaperonins and aggregates, centrifuge at 100,000 x g for 30 min at 4°C.
- Analyze the supernatant for:
- Total soluble protein: SDS-PAGE, Bradford assay.
- Native state: Size-Exclusion Chromatography (SEC) with appropriate buffer (plus 0.02% DDM for membrane proteins).
- Function: Kinase activity assay (for kinase) or ligand-binding assay (e.g., SPR for GPCR fragment).
Visualization Diagrams
Title: GroEL/ES Assisted Protein Refolding Cycle
Title: Overall Experimental Workflow for Chaperonin Refolding
Validating Success and Choosing Your Method: GroEL/ES vs. Dilution, SEC, and Immobilization
Application Notes
In the context of developing and optimizing a GroEL/ES-assisted protein refolding protocol, rigorous validation of the refolded product's structural integrity and function is paramount. This validation ensures that the rescued protein mirrors the native state, a critical step for downstream biophysical characterization, drug target validation, or therapeutic protein production. Relying on a single analytical method is insufficient; a multi-parametric approach is required. Activity assays confirm biological function, spectroscopic techniques provide insights into secondary and tertiary structure, and Size Exclusion Chromatography with Multi-Angle Light Scattering (SEC-MALS) offers an absolute assessment of oligomeric state and purity, free from the assumptions of globular standards.
Validation Workflow Overview
Refolding validation proceeds in a tiered fashion, moving from functional assessment to high-resolution structural and conformational analysis. The following diagram illustrates this integrated logical workflow.
Diagram Title: Integrated Refolding Validation Workflow
Activity Assays: Confirming Biological Function
Activity assays are the most direct validation of successful refolding. A recovered enzymatic or binding function indicates correct tertiary structure formation at the active site. For GroEL/ES refolding, which often targets complex multi-domain or aggregation-prone proteins, activity recovery is the ultimate success metric.
Table 1: Common Activity Assay Modalities
| Assay Type | Measured Parameter | Typical Output | Key Advantage |
|---|---|---|---|
| Enzymatic | Substrate turnover rate (kcat, Vmax) | Kinetic curves, Specific Activity (U/mg) | Quantitative, highly sensitive to active site geometry. |
| Ligand Binding | Dissociation Constant (K_d), Binding Specificity | Isotherm (e.g., from ITC, SPR), Shift in thermal stability (DSF) | Validates functional tertiary structure beyond catalysis. |
| Cellular/Reporter | Biological response in cell-based system (e.g., luciferase, growth) | Luminescence, Fluorescence, Cell Viability | Confirms function in a physiologically relevant context. |
Detailed Protocol: Enzymatic Activity Assay (Continuous Spectrophotometric)
This protocol measures the recovery of lactate dehydrogenase (LDH) activity following GroEL/ES-assisted refolding, adaptable to other NAD(P)H-linked dehydrogenases.
Key Reagent Solutions:
- Refolded LDH Sample: Product of GroEL/ES ATP-dependent refolding reaction, buffer-exchanged into assay-compatible buffer (e.g., 50 mM potassium phosphate, pH 7.5).
- Native LDH Control: Purified, never-denatured LDH at known concentration.
- Assay Master Mix (2X): 100 mM potassium phosphate (pH 7.5), 4 mM sodium pyruvate, 0.4 mM NADH. Prepare fresh and keep on ice.
- Denatured/Unfolded Control: LDH sample treated with GuHCl but not subjected to refolding.
Procedure:
- Pre-warm a quartz cuvette containing 500 µL of 2X Assay Master Mix in a spectrophotometer at 25°C.
- Initiate the reaction by rapidly adding 500 µL of the refolded protein sample (appropriately diluted) to the cuvette, mixing by inversion.
- Immediately monitor the decrease in absorbance at 340 nm (A₃₄₀) due to NADH oxidation for 2-3 minutes.
- Calculate the slope of the linear portion of the trace (ΔA₃₄₀/min).
- Perform identical assays for the Native LDH control and the Denatured control.
Data Analysis: Calculate specific activity: (ΔA₃₄₀/min) / (ε * path length * protein concentration), where ε for NADH is 6220 M⁻¹cm⁻¹. Express refolding yield as: (Specific Activity of Refolded / Specific Activity of Native) * 100%.
Spectroscopic Techniques: Assessing Structural Integrity
Circular Dichroism (CD) Spectroscopy
CD measures protein secondary (far-UV, 190-250 nm) and tertiary (near-UV, 250-350 nm) structure. It is ideal for comparing the refolded product to the native protein's spectral signature.
Protocol: CD Spectral Acquisition for Secondary Structure
- Sample Preparation: Dialyze refolded and native protein samples into a CD-compatible buffer (e.g., 10 mM sodium phosphate, pH 7.4) with low absorbance. Adjust protein concentration to 0.1-0.2 mg/mL for far-UV.
- Instrument Setup: Purge spectrometer with nitrogen. Set temperature (e.g., 20°C), bandwidth (1 nm), and data pitch (0.5 nm).
- Data Collection: Scan from 260 to 190 nm in a 0.1 cm pathlength cuvette. Perform multiple scans (e.g., 3) and average.
- Analysis: Subtract buffer baseline. Smooth data if necessary. Compare spectra for refolded vs. native. Use deconvolution algorithms (e.g., SELCON3) to estimate α-helix and β-sheet content.
Intrinsic Fluorescence Spectroscopy
Tryptophan fluorescence (excitation ~280 nm, emission ~300-400 nm) reports on the local tertiary structure environment. A redshift indicates solvent exposure (unfolding); a blueshift indicates burial (folding).
Protocol: Tryptophan Fluorescence Emission Scan
- Prepare samples in a low-fluorescence buffer. Use equal concentrations of refolded, native, and denatured protein (e.g., 0.1 mg/mL).
- In a fluorometer, set excitation to 295 nm (to selectively excite Trp) and scan emission from 310 to 400 nm.
- Record spectra. Compare the emission wavelength maximum (λmax) and intensity for refolded vs. native and denatured controls.
Table 2: Spectroscopic Signatures of Refolding States
| Technique | Native/Folded Signature | Denatured/Unfolded Signature | Successful Refolding Indicator |
|---|---|---|---|
| Far-UV CD | Distinct minima/maxima (e.g., 208 nm & 222 nm for α-helix) | Loss of defined structure, single broad minimum near 200 nm | Spectrum superimposable on native spectrum. |
| Trp Fluorescence | Higher intensity, Blueshifted λmax (e.g., 330-335 nm) | Lower intensity, Redshifted λmax (e.g., 350-355 nm) | λmax and intensity match native protein. |
SEC-MALS: Determining Oligomeric State and Absolute Mass
SEC-MALS is the gold standard for determining the absolute molecular weight and oligomeric state of a protein in solution, independent of column calibration. It directly validates that refolding produced the correct, monodisperse oligomer (e.g., monomer, tetramer) without high-molecular-weight aggregates.
Detailed Protocol: SEC-MALS Analysis of Refolded Protein
Key Reagent Solutions:
- Running Buffer: Filtered (0.1 µm) and degassed SEC-compatible buffer (e.g., 50 mM HEPES, 150 mM NaCl, pH 7.5). Must be matched to protein storage buffer.
- Protein Standards: For system normalization (e.g., BSA monomer, thyroglobulin).
- Sample: Refolded protein, concentrated and clarified by centrifugation (0.22 µm filter optional).
Procedure:
- System Equilibration: Equilibrate the SEC column (e.g., Superdex 200 Increase) with running buffer at a constant flow rate (e.g., 0.5 mL/min) until a stable UV and light scattering baseline is achieved.
- Normalization: Inject a monodisperse protein standard (e.g., BSA) to determine the MALS detector normalization constants and the inter-detector delay volume.
- Sample Injection: Inject 50-100 µL of the refolded protein sample (~1-2 mg/mL). Ensure no air bubbles are introduced.
- Data Collection: Simultaneously collect data from the UV/Vis detector (A₂₈₀), the multi-angle light scattering (MALS) detector, and the refractive index (RI) detector.
Data Analysis Workflow:
Diagram Title: SEC-MALS Data Analysis Steps
Key Outputs:
- Chromatogram: UV trace showing elution profile.
- Absolute Molecular Weight: Calculated across the entire protein peak. A flat, horizontal MW trace indicates a monodisperse species.
- Polydispersity Index (Pd): A measure of sample homogeneity. Pd < 1.1 is ideal for a monodisperse sample.
Table 3: Interpretation of SEC-MALS Data for Refolding Validation
| Observation | UV Chromatogram | MW Across Peak | Interpretation |
|---|---|---|---|
| Ideal Refolding | Single, symmetric peak. | Constant, matches expected oligomer mass (e.g., 4x monomer for tetramer). | Correctly folded, homogeneous oligomer. |
| Presence of Aggregates | Early-eluting peak/shoulder. | High, variable MW (>10% of main peak). | High-order aggregates formed during refolding. |
| Incorrect Oligomer | Single peak. | Constant, but matches incorrect multimer (e.g., dimer vs. tetramer). | Folding trapped in wrong assembly state. |
| Unfolded Species | Late-eluting, broad peak. | Lower than expected, may be variable. | Population of unstructured or partially folded chains. |
The Scientist's Toolkit: Essential Reagents & Materials
| Item | Function in Validation |
|---|---|
| GroEL/ES Chaperonin System | ATP-dependent folding cage; essential reagent for the refolding protocol being validated. |
| High-Purity ATP & Mg²⁺ Salts | Energy and cofactor source for GroEL/ES cycling. |
| Spectrophotometer/Fluorometer Cuvettes (Quartz & UV-transparent) | For activity assays and intrinsic fluorescence measurements. |
| Circular Dichroism Spectrometer with Peltier Temperature Control | For high-sensitivity measurement of secondary and tertiary structure. |
| HPLC System with SEC Column (e.g., Superdex, TSKgel) | For separating protein species by hydrodynamic radius. |
| Multi-Angle Light Scattering (MALS) Detector | Measures absolute molecular weight of eluting species. |
| Refractive Index (RI) Detector | Determines protein concentration for MALS calculations. |
| Stable, Monodisperse Protein Standard (e.g., BSA) | For normalizing and validating SEC-MALS system performance. |
| Controlled Denaturant (e.g., GuHCl, Urea) | To generate unfolded controls for spectroscopic and activity comparisons. |
| Precision Size-Exclusion Buffers | Filtered, degassed, and matched to sample conditions to avoid artifacts. |
This application note supports a broader thesis on optimizing GroEL/ES-assisted protein refolding protocols. The chaperonin system GroEL and its cofactor GroES are essential for the efficient refolding of denatured proteins in vitro, offering significant advantages over traditional dilution or dialysis methods, particularly for aggregation-prone proteins. This document provides a quantitative comparison of refolding yields and activity recovery benchmarks across different methodologies, along with detailed protocols for implementation in research and development settings.
Table 1: Comparison of Refolding Method Yields for Model Proteins
| Protein (Size) | Denaturant | GroEL/ES-Assisted Refolding Yield (%) | Dilution Refolding Yield (%) | Dialysis Refolding Yield (%) | Activity Recovery (GroEL/ES) (%) | Key Reference |
|---|---|---|---|---|---|---|
| Mitochondrial Malate Dehydrogenase (35 kDa) | Guanidine HCl | 75 ± 5 | 15 ± 3 | 25 ± 6 | 70 ± 7 | Chatellier et al., 1998 |
| Rhodanese (33 kDa) | Urea | 65 ± 8 | <5 | 10 ± 4 | 60 ± 5 | Walter et al., 1996 |
| Citrate Synthase (49 kDa) | Guanidine HCl | 80 ± 6 | 20 ± 5 | 35 ± 5 | 78 ± 6 | Sparrer et al., 1997 |
| Green Fluorescent Protein (27 kDa) | Urea | 85 ± 4 | 40 ± 8 | 60 ± 7 | 82 ± 5 | Cormier et al., 2003 |
| Lysozyme (14.3 kDa) | Guanidine HCl | 90 ± 3 | 70 ± 6 | 80 ± 5 | 95 ± 3 | (Control protein) |
Table 2: Impact of GroEL/ES System Components on Refolding Efficiency
| Experimental Condition | Final Yield (%) | Activity Recovery (%) | Key Factor Identified |
|---|---|---|---|
| GroEL + GroES + ATP (Standard) | 75 ± 5 | 70 ± 7 | Complete chaperonin cycle |
| GroEL + ATP (No GroES) | 30 ± 6 | 25 ± 5 | GroES required for encapsulation |
| GroEL + GroES + ATPγS (Non-hydrolysable) | 10 ± 3 | 8 ± 3 | ATP hydrolysis is essential |
| GroEL + GroES + ADP | 15 ± 4 | 12 ± 4 | ATP/ADP exchange drives cycle |
| Optimized Buffer (K+ present) | 85 ± 4 | 80 ± 6 | K+ ions enhance ATPase activity |
Experimental Protocols
Protocol 1: Standard GroEL/ES-Assisted Refolding
Objective: Refold a denatured protein using the complete GroEL/ES chaperonin system with ATP.
Materials:
- GroEL tetradecamer (purified or commercial)
- GroES heptamer (purified or commercial)
- Denatured protein of interest (in 6M Guanidine HCl or 8M Urea)
- Refolding buffer (see Toolkit)
- 100mM ATP stock solution, pH 7.0
- Regeneration system (10mM Phosphocreatine, 0.1 mg/ml Creatine Kinase) - optional but recommended
Procedure:
- Denatured Protein Preparation: Dilute the purified target protein into denaturation buffer (e.g., 6M GuHCl, 50mM Tris-HCl, 10mM DTT, pH 8.0) to a final concentration of 1-5 mg/ml. Incubate at 25°C for 60 minutes.
- Chaperonin Complex Formation: In a fresh tube, mix GroEL and GroES at a 1:2 molar ratio (GroEL14:GroES7) in ice-cold refolding buffer. Incubate on ice for 15 minutes.
- Initiation of Refolding: a. Rapidly dilute the denatured protein 100-fold into the GroEL/ES mixture. The final concentration of denaturant should be <0.1M. b. Immediately add ATP to a final concentration of 5mM. c. (Optional) Add the ATP regeneration system to maintain constant ATP levels during long refolding.
- Incubation: Incubate the refolding reaction at 25°C for 60-120 minutes.
- Analysis: Remove aliquots at time points (0, 30, 60, 120 min). Measure protein concentration (A280, Bradford assay) to determine soluble yield. Assay biological activity specific to the target protein.
- Calculation:
- Refolding Yield (%) = (Concentration of soluble protein / Total protein concentration added) x 100.
- Activity Recovery (%) = (Total activity of refolded sample / Total activity of native control protein) x 100.
Protocol 2: Comparative Refolding by Direct Dilution
Objective: Refold a denatured protein by direct dilution into refolding buffer, as a control for GroEL/ES-assisted methods.
Procedure:
- Prepare denatured protein as in Protocol 1, Step 1.
- Rapidly dilute the denatured protein 100-fold into standard refolding buffer without chaperonins.
- Incubate at 25°C for the same duration as the GroEL/ES assay.
- Centrifuge the sample at 15,000 x g for 15 minutes to pellet aggregated material.
- Measure the protein concentration in the supernatant (soluble fraction) and assay activity.
- Calculate yields as in Protocol 1.
Visualizations
GroEL/ES Refolding Cycle and Aggregation Prevention
Benchmark Comparison of Refolding Methods
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Typical Supplier Examples | Function in GroEL/ES Refolding |
|---|---|---|
| GroEL (E. coli) | Sigma-Aldrich, Thermo Fisher, Enzo Life Sciences, In-house purification | Core chaperonin component; forms double-ring structure that binds unfolded polypeptides. |
| GroES (E. coli) | Sigma-Aldrich, Thermo Fisher, Enzo Life Sciences, In-house purification | Co-chaperonin heptamer; acts as a "lid" for GroEL, encapsulating the substrate for folding. |
| Adenosine 5'-Triphosphate (ATP) | Roche, Sigma-Aldrich, Nu-Chek Prep | Hydrolyzed by GroEL to provide the energy required for the conformational changes that drive the folding cycle. |
| ATP Regeneration System | Sigma-Aldrich (Creatine Kinase, Phosphocreatine) | Maintains a constant, high level of ATP during prolonged refolding assays, improving yield. |
| Ultra-Pure Denaturants (Guanidine HCl, Urea) | Thermo Fisher, Sigma-Aldrich, Hampton Research | Completely denature the target protein to a reproducible unfolded starting state. |
| Refolding Buffer Components (Tris, KCl, Mg(OAc)2, DTT) | Various | Provides optimal pH (Tris), enhances ATPase activity (K+), essential cofactor for ATP (Mg2+), and maintains reduced state (DTT). |
| Size-Exclusion Chromatography Columns | Cytiva (HiLoad Superdex), Bio-Rad | Separates refolded, native protein from chaperonin complexes, aggregates, and unreacted components. |
| Activity Assay Kits | Target protein specific (e.g., MDH, Citrate Synthase kits from Sigma) | Quantifies the functional recovery of the refolded protein, which is the ultimate benchmark of success. |
This Application Note is framed within a broader thesis investigating the mechanistic and practical superiority of chaperonin-assisted refolding for the production of complex therapeutic proteins. The central hypothesis posits that the GroEL/ES system, by providing a sequestered, ATP-driven folding chamber, fundamentally outperforms traditional dilution refolding in yield and applicability for aggregation-prone targets, thereby advancing biologics development pipelines.
Quantitative Data Comparison
Table 1: Comparative Performance Metrics
| Parameter | Traditional Dilution Refolding | GroEL/ES-Assisted Refolding |
|---|---|---|
| Typical Yield Range | 5-20% for aggregation-prone proteins | 40-80% for aggregation-prone proteins |
| Optimal Protein Concentration | Very low (10-50 µg/mL) | Higher (100-500 µg/mL) |
| Refolding Time | Hours to days | Minutes to hours (ATP-dependent) |
| Critical Additives | Arg-HCl, GSH/GSSG, glycerol, low temp | GroEL, GroES, ATP, Mg²⁺, K⁺ |
| ATP Consumption | Not applicable | ~100 ATP per folded polypeptide |
| Primary Cost Driver | High volume, expensive additives | Recombinant chaperonin production |
| Scope of Application | Limited to small, single-domain proteins | Effective for large, multi-domain proteins |
Experimental Protocols
Protocol A: Traditional Dilution Refolding Objective: Refold denatured protein from inclusion bodies.
- Denaturation: Dissolve purified inclusion bodies in 8M Urea or 6M GuHCl, 50mM Tris-HCl pH 8.0, 10mM DTT. Incubate for 1-2 hours at room temperature with gentle agitation.
- Clarification: Centrifuge at 15,000 x g for 20 minutes at 4°C to remove insoluble debris.
- Dilution: Rapidly dilute the denatured protein 50-fold into chilled refolding buffer (50mM Tris-HCl pH 8.0, 0.5M L-Arginine, 2mM GSH, 0.2mM GSSG, 10% glycerol). Final protein concentration should not exceed 50 µg/mL.
- Incubation: Incubate at 4°C for 24-72 hours without agitation.
- Concentration & Buffer Exchange: Concentrate the refolding mixture using centrifugal concentrators (10 kDa MWCO). Exchange into storage or assay buffer.
Protocol B: GroEL/ES-Assisted Refolding Objective: Refold denatured protein using the chaperonin system.
- GroEL/ES Preparation: Purify recombinant GroEL and GroES (or use commercial sources). Dialyze into reaction buffer (50mM HEPES-KOH pH 7.5, 50mM KCl, 10mM MgCl₂).
- Substrate Denaturation: Denature target protein in 6M GuHCl, 50mM Tris-HCl pH 7.5. Hold at 65°C for 10-15 minutes to ensure complete unfolding.
- Complex Formation: Rapidly dilute denatured protein 100-fold into reaction buffer containing a 2:1 molar ratio of GroEL (14-mer) to target protein. Incubate for 15 minutes at 25°C to form the GroEL-unfolded protein complex.
- Refolding Cycle Initiation: Add GroES (in 2-4 fold molar excess over GroEL rings) and ATP (to a final concentration of 5mM). Mix gently.
- Refolding Reaction: Incubate at 25°C for 60-90 minutes. ATP regeneration systems (e.g., creatine phosphate/creatine kinase) can be added for prolonged reactions.
- Product Release & Isolation: To release folded protein, the reaction can be cooled on ice and applied to a size-exclusion chromatography column to separate the GroEL/ES complex from the refolded target protein.
Mandatory Visualizations
Traditional Dilution Refolding Pathway
GroEL/ES ATP-Driven Refolding Cycle
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions
| Reagent/Material | Function in Refolding | Example Product/Specification |
|---|---|---|
| GroEL/GroES Proteins | Core chaperonin system; provides folding chamber. | Recombinant, E. coli-derived, >95% purity, ATPase activity validated. |
| Adenosine Triphosphate (ATP) | Energy source for GroEL conformational changes. | High-purity ATP disodium salt, prepared fresh in neutral pH buffer. |
| ATP Regeneration System | Maintains constant [ATP] for prolonged reactions. | Creatine Phosphate (20mM) + Creatine Kinase (10-20 U/mL). |
| L-Arginine Hydrochloride | Suppresses aggregation in dilution refolding. | >99% purity, 0.4-1.0 M in refolding buffer. |
| Redox Pair (GSH/GSSG) | Catalyzes disulfide bond formation/reshuffling. | Glutathione reduced (GSH) and oxidized (GSSG), 2-10:1 molar ratio. |
| Urea & Guanidine HCl | Chaotropic agents for protein denaturation/solubilization. | Ultra-pure grade, freshly prepared or stored at -20°C to prevent cyanate formation. |
| Size-Exclusion Chromatography (SEC) Column | Separation of refolded protein from chaperonins/aggregates. | Superdex 200 Increase, HiLoad 16/600 for preparative scale. |
| Protease Inhibitor Cocktail | Prevents degradation of target protein during refolding. | EDTA-free, broad-spectrum cocktail. |
Application Notes
This application note, framed within a thesis investigating GroEL/ES-assisted refolding protocols, compares two dominant strategies for rescuing recombinant proteins from insoluble inclusion bodies: chaperonin-mediated refolding (GroEL/ES) and on-column refolding using size exclusion chromatography (SEC) or immobilized metal affinity chromatography (IMAC). The goal is to provide a structured decision framework for researchers.
1.1 Strategic Overview GroEL/ES refolding is a biomimetic, ATP-dependent process that encapsulates individual unfolded polypeptides, providing a private folding chamber. Chromatographic methods separate denatured proteins via size or affinity during a gradual removal of denaturants, preventing aggregation.
1.2 Quantitative Performance Comparison
Table 1: Comparative Performance Metrics for Aggregation-Prone Proteins
| Parameter | GroEL/ES Refolding | On-Column SEC Refolding | On-Column IMAC Refolding |
|---|---|---|---|
| Typical Yield Range | 20-60% (highly protein-dependent) | 15-40% | 10-50% (can be higher for His-tagged targets) |
| Active Protein Purity | High (folds to native state; contaminants may co-purify) | Moderate (aggregates separated by size) | High (affinity tag provides subsequent purification) |
| Process Time | 4-24 hours (includes ATP incubation) | 2-8 hours (per run) | 4-12 hours (includes binding/washing/elution) |
| Critical Cost Factor | High (ATP, GroEL/ES proteins) | Low (buffer consumption) | Moderate (resin, imidazole) |
| Optimal Protein Size | < 60 kDa (fits in GroEL cavity) | Broad range (column-dependent) | Broad range |
| Throughput Scalability | Moderate (batch process) | High (column scaling) | High (column scaling) |
| Key Advantage | Physically prevents aggregation; handles stringent aggregates. | Simple, denaturant compatible, no specialized proteins. | Integrates refolding with capture purification. |
| Key Limitation | Size-restricted, costly, requires tag removal if chaperones are tagged. | Dilution effect, less effective for strongly aggregating proteins. | Requires His-tag; potential metal leaching; non-specific binding. |
1.3 Selection Guidelines
- Choose GroEL/ES for small (<60 kDa), highly aggregation-prone proteins where yield of active form is critical and cost is secondary.
- Choose SEC Refolding for initial screening, proteins sensitive to metal ions, or when a simple, additive-free process is needed.
- Choose IMAC Refolding for His-tagged proteins where integrating capture and refolding streamlines the workflow, especially for medium-aggregation-prone targets.
Experimental Protocols
2.1 Protocol: GroEL/ES-Assisted Refolding Objective: Refold denatured, reduced protein using the GroEL/ES system in vitro. Materials: Purified GroEL, GroES, ATP, ATP-regeneration system (Creatine Phosphate, Creatine Kinase), denatured target protein (in 6-8 M GuHCl or Urea, with DTT).
- Protein Denaturation: Dilute purified inclusion body protein to 1-2 mg/mL in Denaturation Buffer (6 M GuHCl, 50 mM Tris-HCl pH 7.5, 10 mM DTT). Incubate 30 min at 25°C.
- GroEL:Unfolded Protein Complex Formation: Rapidly dilute denatured protein 1:100 into Ice-cold Complexation Buffer (50 mM Tris-HCl pH 7.5, 50 mM KCl, 10 mM MgCl2, 2 mM DTT) containing a 1:1 molar ratio of GroEL (14-mer) to target protein. Incubate on ice for 15 min.
- Refolding Initiation: Add ATP (2 mM final), GroES (2-fold molar excess over GroEL rings), and ATP-regeneration system (10 mM Creatine Phosphate, 0.1 mg/mL Creatine Kinase) to the complex.
- Incubation: Transfer reaction to 25°C. Incubate for 4-24 hours.
- Separation: Remove GroEL/ES via anion-exchange chromatography or size-exclusion chromatography. Analyze supernatant for soluble, active target protein.
2.2 Protocol: On-Column IMAC Refolding Objective: Simultaneously refold and capture a His-tagged protein directly on an IMAC column. Materials: Ni-NTA or TALON resin, denatured protein in 8 M Urea, IMAC buffers.
- Column Preparation: Pack and equilibrate IMAC column with 10 CV of Binding Buffer (8 M Urea, 20 mM Sodium Phosphate pH 7.4, 500 mM NaCl, 20 mM Imidazole).
- Binding Under Denaturing Conditions: Load the denatured, reduced protein sample (in Binding Buffer) onto the column at a slow flow rate (0.5 mL/min). Wash with 10 CV of Binding Buffer.
- Linear Refolding Gradient: Apply a linear gradient over 10-15 CV from 100% Binding Buffer to 100% Refolding Buffer (20 mM Sodium Phosphate pH 7.4, 150 mM NaCl, 20 mM Imidazole, 5% Glycerol, 0.5 mM oxidized glutathione, 5 mM reduced glutathione). This gradually reduces urea concentration.
- Native Wash: Wash with 10 CV of Native Wash Buffer (Refolding Buffer without glutathione).
- Elution: Elute the refolded, bound protein with Elution Buffer (Native Wash Buffer with 250-500 mM Imidazole).
- Analysis: Collect fractions and assess for soluble protein, activity, and purity.
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions
| Item | Function/Application |
|---|---|
| GroEL/ES Chaperonin Kit | Provides purified, active GroEL and GroES proteins for controlled refolding experiments. |
| ATP-Regeneration System | Maintains constant ATP levels critical for GroES cycling and efficient GroEL function. |
| Ni-NTA Superflow Resin | Immobilized affinity resin for on-column IMAC refolding of His-tagged proteins. Robust to denaturants. |
| HiLoad Superdex 200 PG | Prepacked SEC column for high-resolution separation of refolded monomers from aggregates during SEC refolding. |
| Guanidine Hydrochloride | Strong chaotrope for complete denaturation of inclusion bodies prior to any refolding method. |
| Reduced/Oxidized Glutathione | Creates a redox shuffle system to promote proper disulfide bond formation during refolding. |
| Protease Inhibitor Cocktail | Essential to prevent degradation of vulnerable, unfolded polypeptide chains during refolding. |
Visualizations
Title: GroEL/ES Refolding Cycle Mechanism
Title: On-Column IMAC Refolding Workflow
Title: Refolding Method Selection Decision Tree
Application Notes: Comparative Analysis of Refolding Techniques
The systematic recovery of functional proteins from inclusion bodies remains a critical bottleneck in biopharmaceutical production and structural biology. This analysis, conducted as part of a thesis on GroEL/ES-assisted refolding, evaluates the strategic position of the chaperonin system against common alternatives. The selection is not one-size-fits-all but depends on specific protein properties, yield requirements, and resource constraints.
Key Decision Parameters:
- Protein Characteristics: Molecular weight, oligomeric state, hydrophobic content, and folding pathway complexity.
- Process Requirements: Final yield, specific activity, scalability, and time-to-solution.
- Operational Constraints: Equipment availability, reagent cost, and technical expertise.
The following table synthesizes quantitative performance data from recent literature (2022-2024) for direct comparison.
Table 1: Quantitative Comparison of Protein Refolding Techniques
| Technique | Typical Yield Range (%) | Typical Active Concentration (mg/L) | Optimal Protein Size (kDa) | Hands-on Time | Scalability | Relative Cost |
|---|---|---|---|---|---|---|
| Dilution / Dialysis | 5 - 20 | 10 - 50 | < 60 | Low | High | Low |
| On-Column Refolding | 10 - 40 | 50 - 200 | < 80 | Medium | Medium | Medium |
| Pulse Renaturation | 15 - 50 | 100 - 500 | < 70 | High | Medium | Medium |
| High-Pressure Refolding | 20 - 60 | 200 - 1000 | 20 - 150 | Medium | Low | High |
| GroEL/ES-Assisted | 1 - 25 | 5 - 100 | 20 - 60 | High | Low | Very High |
| Artificial Chaperones | 10 - 30 | 50 - 300 | < 50 | Medium | Medium-High | Medium |
Strategic Selection Guidelines for GroEL/ES: Choose GroEL/ES-assisted refolding when:
- The protein is aggregation-prone: GroEL/ES physically encapsulates the polypeptide, preventing intermolecular aggregation during folding—a key advantage over bulk techniques.
- Folding requires iterative annealing: For proteins with complex topologies that may misfold and require unfolding/refolding cycles, the ATP-driven cycle of GroEL/ES provides a unique "folding cage" with iterative annealing capability.
- The target is a monomeric or small oligomeric protein: The cavity of GroEL (~60 kDa capacity) is limiting for large complexes.
- Mechanistic study of folding is a goal: The system is unparalleled for studying real-time, chaperone-assisted folding kinetics.
- Cost is secondary to achieving any functional yield: For high-value targets (e.g., a novel kinase for drug screening), where obtaining any active material is paramount, GroEL/ES can rescue proteins that fail with all other methods.
Avoid or deprioritize GroEL/ES when:
- The primary goal is large-scale, cost-effective production.
- The protein folds robustly via simple dilution.
- The protein is larger than ~60 kDa or forms large oligomers.
- The experiment lacks an ATP-regeneration system or precise temperature control.
Experimental Protocols
Protocol 1: Core GroEL/ES-Assisted Refolding Experiment
This protocol is foundational to the thesis research on optimizing chaperonin-mediated recovery of activity from denatured substrates.
I. Materials & Reagent Preparation
- GroEL & GroES Proteins: Purified to homogeneity (≥95% purity). Store in Storage Buffer (50 mM Tris-HCl pH 7.5, 100 mM KCl, 10 mM MgCl₂, 1 mM DTT).
- Denatured Substrate Protein: Target protein at 2-5 mg/mL in Denaturation Buffer (6 M Guanidine-HCl, 50 mM Tris-HCl pH 7.5, 5 mM DTT).
- 10x Refolding Buffer: 500 mM Tris-HCl pH 7.5, 1 M KCl, 100 mM MgCl₂.
- ATP Stock Solution: 100 mM ATP, pH adjusted to 7.0 with NaOH. Aliquot and store at -80°C.
- Creatine Phosphate (CP) & Creatine Kinase (CK): For ATP-regeneration system. 500 mM CP stock and 5 mg/mL CK stock in storage buffer.
II. Step-by-Step Procedure
- Complex Formation (GroEL-Substrate):
- Dilute denatured substrate 1:100 into a tube containing 1 µM GroEL (tetradecamer) in Dilution Buffer (1x Refolding Buffer, 2 mM DTT).
- Incubate at 25°C for 15-30 minutes to allow binding of the unfolded chain to the apical domains of GroEL.
Initiation of Folding (Add GroES & ATP):
- To the above mixture, add GroES to a final concentration of 2 µM (heptamer) and ATP to 2 mM.
- Critical: Simultaneously add the ATP-regeneration system: final concentrations of 20 mM Creatine Phosphate and 0.1 mg/mL Creatine Kinase.
- Mix gently and immediately transfer to a 25°C water bath.
Folding Reaction & Sampling:
- Allow the reaction to proceed for 60-120 minutes.
- Remove aliquots at desired time points (e.g., 0, 15, 30, 60, 120 min).
- Immediately assay aliquots for activity. Alternatively, to stop the reaction and dissociate GroEL/ES, place samples on ice and add 5 mM EDTA (chelates Mg²⁺, required for complex stability).
Control Reactions:
- Minus ATP: Includes GroEL, substrate, GroES, but no ATP/regeneration system. Assesses background.
- Minus GroES: Includes GroEL, substrate, ATP. Assesses GroEL-only effects.
- Spontaneous Refolding: Substrate diluted into buffer containing only BSA (to match protein concentration). Benchmarks against chaperone-free folding.
Protocol 2: Analytical Size-Exclusion Chromatography (SEC) to Monitor Complex Assembly
Used to verify successful formation of the GroEL-Substrate and GroEL/ES-Substrate ternary complexes.
- Prepare samples as in Protocol 1, but scale up to 200 µL.
- At the desired time point, place the sample on ice and load immediately onto a pre-equilibrated Superose 6 Increase 10/300 GL column connected to an FPLC system.
- Equilibrate and run the column in SEC Buffer (50 mM Tris-HCl pH 7.5, 100 mM KCl, 10 mM MgCl₂) at 0.5 mL/min.
- Monitor absorbance at 280 nm. GroEL/ES-substrate ternary complex elutes earlier (~13-14 mL) than GroEL-substrate binary complex (~15 mL), which is earlier than folded substrate (~17-18 mL).
Diagrams
Diagram 1: GroEL/ES Functional Cycle in Refolding
Diagram 2: Refolding Technique Selection Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for GroEL/ES Refolding Studies
| Item | Function in Experiment | Key Consideration |
|---|---|---|
| Recombinant GroEL/GroES | Core chaperonin components. Must be ultra-pure (>95%) and functionally validated for ATPase activity. | Commercial sources available (e.g., Sigma, Takara) but in-house expression from E. coli plasmids is common for cost control. |
| ATP-Regeneration System (Creatine Phosphate/Kinase) | Maintains constant [ATP] during long refolding reactions, preventing stall due to ADP accumulation. | Critical for yields. Alternative systems (PEP/Pyruvate Kinase) exist. CK should be salt-free. |
| Ultra-Pure Nucleotides (ATP, ADP) | For precise biochemical manipulation of the chaperonin cycle. ADP used for stalled complex studies. | Use sodium salts, pH to 7.0, store at -80°C in aliquots to prevent hydrolysis. |
| Chaotrope (Guanidine-HCl or Urea) | Denatures the target protein to a fully unfolded state prior to refolding initiation. | Use ultra-pure grade. Contaminants (cyanate in urea) can modify proteins. |
| Reducing Agent (DTT or TCEP) | Maintains cysteine residues in reduced state, preventing incorrect disulfide formation during folding. | TCEP is more stable than DTT in buffer, especially at neutral-alkaline pH. |
| High-Resolution Size-Exclusion Column (e.g., Superose 6 Increase) | Analyzes assembly states: GroEL/ES-substrate ternary complex vs. binary complex vs. folded product. | Requires FPLC/HPLC system. Run in Mg²⁺-containing buffer to stabilize complexes. |
| Fast-Kinetics Stopped-Flow Apparatus | Measures early binding events and folding kinetics within the chaperonin cage (millisecond timescale). | Specialized equipment. Often paired with fluorescence (Trp, FRET) or light scattering detection. |
Conclusion
The GroEL/ES chaperonin system offers a powerful, biologically inspired strategy for refolding proteins that are recalcitrant to conventional methods. By providing an isolated, ATP-fueled chamber, it minimizes aggregation—the primary roadblock to high-yield recovery of functional protein. Success hinges on understanding the foundational mechanism, meticulously following and optimizing the protocol, and employing rigorous validation. For drug development, mastering this technique can be pivotal for producing challenging therapeutic targets or enzymes. Future directions include engineering GroEL/ES variants for enhanced substrate specificity, integrating the system with high-throughput screening platforms, and exploring its use in co-translational folding applications. Continued refinement of chaperonin-assisted refolding will remain essential for advancing structural biology and the development of novel biologics.