Chaperone-Mediated Autophagy (CMA): The Essential Protein Quality Control Pathway in Health and Disease
This article provides a comprehensive overview of Chaperone-Mediated Autophagy (CMA), a selective lysosomal degradation pathway crucial for cellular protein quality control.
Chaperone-Mediated Autophagy (CMA): The Essential Protein Quality Control Pathway in Health and Disease
Abstract
This article provides a comprehensive overview of Chaperone-Mediated Autophagy (CMA), a selective lysosomal degradation pathway crucial for cellular protein quality control. Targeted at researchers and drug development professionals, it covers the molecular mechanism of CMA, from substrate recognition via HSC70 to LAMP2A-mediated lysosomal translocation. It details cutting-edge methodologies for CMA monitoring and modulation, explores common experimental challenges and optimization strategies, and validates CMA's role through comparative analysis with other proteolytic systems (ubiquitin-proteasome, macroautophagy). The review synthesizes CMA's implications in neurodegeneration, cancer, and aging, highlighting its potential as a therapeutic target.
Decoding Chaperone-Mediated Autophagy: The Molecular Mechanism of Selective Protein Clearance
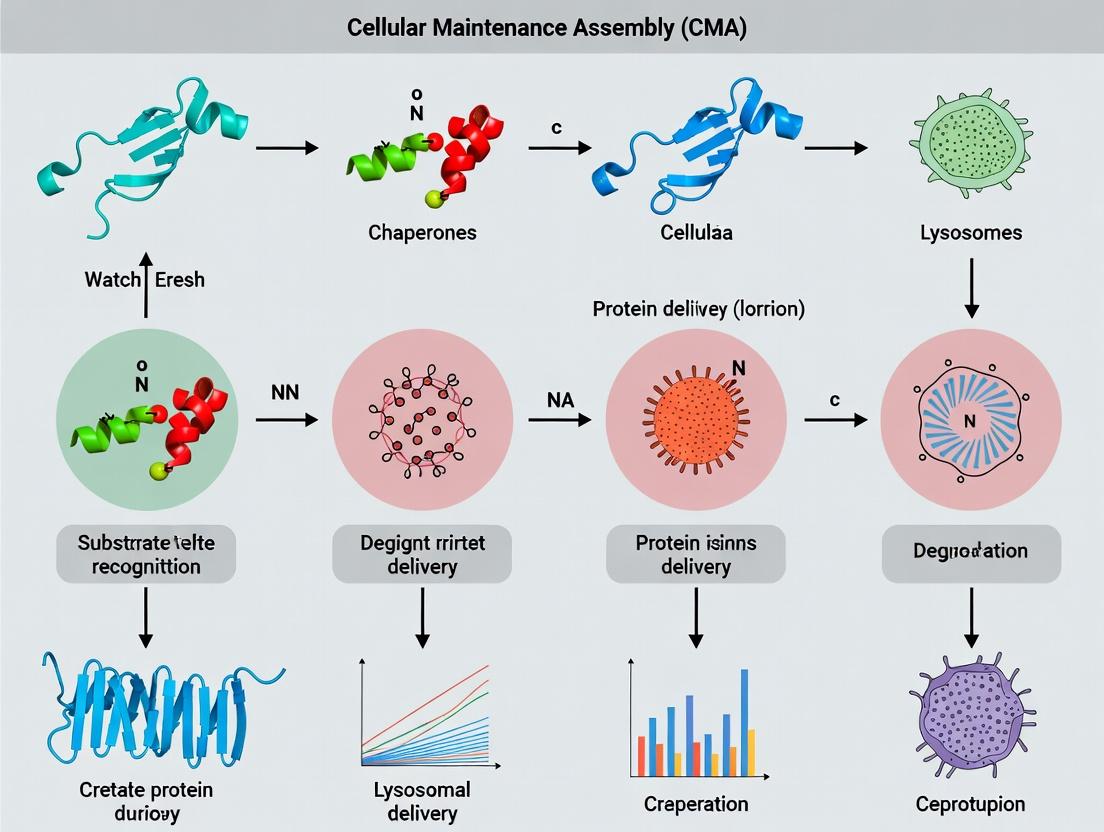
1. Introduction Chaperone-Mediated Autophagy (CMA) is a selective lysosomal degradation pathway integral to cellular protein quality control. Unlike macroautophagy, CMA does not involve vesicular trafficking. Instead, it directly translocates individual cytosolic proteins bearing a specific pentapeptide motif (KFERQ-like) across the lysosomal membrane for degradation. This whitepaper delineates CMA's mechanism, experimental analysis, and its context within cellular proteostasis, providing a technical guide for researchers and drug development professionals.
2. Core Mechanism & Molecular Players CMA involves a series of discrete, regulated steps: substrate recognition, lysosome binding, substrate unfolding, and translocation.
Table 1: Core Components of the CMA Pathway
| Component | Protein/Complex | Primary Function |
|---|---|---|
| Substrate Recognition | Hsc70 (HSPA8) & Co-chaperones | Binds KFERQ motif in cytosolic substrates. |
| Lysosomal Receptor | LAMP2A | Single-span membrane protein; receptor for CMA substrate/chaperone complex. |
| Translocation Complex | LAMP2A Multimer | Forms a 700 kDa complex at the lysosomal membrane; provides the translocation channel. |
| Luminal Chaperone | Lys-Hsc70 (HSPA8) | Resident lysosomal Hsc70; provides inward pulling force for substrate translocation. |
| Regulatory Protein | GFAP (Glial Fibrillary Acidic Protein) | Stabilizes the LAMP2A multimeric complex. |
| Regulatory Protein | EEF1A1 (Elongation Factor 1 Alpha 1) | Binds lysosomal membrane; promotes disassembly of LAMP2A translocon post-translocation. |
Diagram 1: Stepwise mechanism of CMA substrate processing.
3. Quantitative Analysis of CMA Activity CMA flux can be measured using several quantitative approaches. Key metrics include rates of protein degradation, LAMP2A levels, and lysosomal association of substrates.
Table 2: Quantitative Metrics for CMA Assessment
| Assay | Measured Parameter | Typical Experimental Output | Significance |
|---|---|---|---|
| Long-lived Protein Degradation | % Radioactivity released from cells pre-labeled with ³H-Leucine/¹⁴C-Valine in presence of lysosomal inhibitors. | CMA contributes ~30% of total lysosomal degradation under basal conditions in mouse fibroblasts. | Measures overall lysosomal/CMA flux. |
| Photoactivatable KFERQ-Reporters (e.g., PA-mKeima-KFERQ) | Lysosomal delivery (acidic, 580 nm signal) vs. cytosolic localization (neutral pH, 458 nm signal) via flow cytometry. | CMA-active cells show a ~3-5 fold increase in 580/458 nm signal ratio upon CMA induction (e.g., serum starvation). | Direct, real-time measurement of CMA substrate delivery. |
| LAMP2A Levels | Protein abundance via immunoblot of lysosomal membranes. | CMA activity correlates with LAMP2A protein, not mRNA. Oxidative stress can increase LAMP2A by 2-3 fold. | Key regulatory node for CMA capacity. |
| Substrate Binding/ Uptake | Isolated lysosomes incubated with purified radiolabeled substrate (e.g., ¹²⁵I-GAPDH). | Specific uptake is KFERQ-dependent, ATP- and lys-Hsc70-dependent, saturable (Km ~2-5 μM). | Measures functional competence of lysosomes for CMA. |
4. Experimental Protocols
4.1. Protocol: Isolation of CMA-Active Lysosomes
- Principle: Fractionate lysosomes from tissues/cells for functional in vitro binding/uptake assays.
- Method:
- Homogenize liver tissue or cells in cold 0.25 M sucrose, 10 mM MOPS (pH 7.3), 1 mM EDTA.
- Perform differential centrifugation: 800g (10 min) to remove nuclei/debris; 20,000g (10 min) to pellet heavy mitochondria/lysosomes.
- Resuspend pellet and layer onto a discontinuous Percoll gradient (e.g., 12%, 18%, 27% in homogenization buffer).
- Centrifuge at 48,000g for 90 min.
- Collect the dense band (CMA-active lysosomes are denser due to LAMP2A enrichment).
- Wash lysosomes by dilution and centrifugation at 20,000g.
4.2. Protocol: In Vitro CMA Substrate Uptake Assay
- Principle: Measure translocation of a canonical CMA substrate into isolated lysosomes.
- Method:
- Substrate Preparation: Purify a model CMA substrate (e.g., GAPDH or RNase A) and radiolabel with ¹²⁵I or fluorescently tag.
- Incubation: Incubate isolated lysosomes (50-100 μg protein) with substrate (5-10 μg) in 3 mg/ml ATP, 10 mg/ml creatine phosphate, 0.1 mg/ml creatine kinase, in uptake buffer (10 mM HEPES pH 7.4, 0.3 M sucrose, 5 mM MgCl₂, 5 mM DTT) for 20 min at 37°C.
- Control: Include parallel samples with lysosomes pretreated with protease (to degrade LAMP2A) or without ATP.
- Termination & Measurement: Place on ice. Treat with Proteinase K to degrade non-internalized substrate. Pellet lysosomes through a sucrose cushion. Measure radioactivity/fluorescence in the pellet via gamma counter or fluorimeter. Specific uptake = Total signal - signal in protease/ATP-depleted controls.
Diagram 2: Workflow for isolating lysosomes and assaying CMA activity.
5. The Scientist's Toolkit: Key Reagent Solutions
Table 3: Essential Research Reagents for CMA Studies
| Reagent/Category | Specific Example(s) | Function in CMA Research |
|---|---|---|
| CMA Reporters | PA-mKeima-KFERQ constructs, KFERQ-Dendra2, GAPDH-GFP. | Live-cell, real-time visualization and quantification of CMA substrate delivery to lysosomes. |
| LAMP2A-Specific Antibodies | Mouse monoclonal (H4B4), Rabbit polyclonal (ab18528). | Detection of LAMP2A protein levels by immunoblot, immunofluorescence, or immunoprecipitation. |
| Chemical Modulators | 6-Aminonicotinamide (6-AN), Torin 1, PI3K Inhibitors (e.g., 3-MA), Vitamin D derivatives. | 6-AN induces oxidative stress & activates CMA; Torin 1 inhibits mTOR to induce macroautophagy; used for comparative studies. |
| Lysosomal Inhibitors | Bafilomycin A1, Chloroquine, Leupeptin/E64d cocktail. | Inhibit lysosomal acidification or protease activity; used to block degradation and measure flux. |
| Validated CMA Substrates | Purified GAPDH, RNase A, IκBα, MEF2D. | Used in in vitro uptake assays with isolated lysosomes to measure CMA functionality. |
| siRNA/shRNA Libraries | siRNAs targeting LAMP2A, Hsc70 (HSPA8), GFAP. | Genetic knockdown to establish necessity of specific components for CMA. |
| CMA-Defective Models | LAMP2A knockout mice, LAMP2A-/- cell lines. | Essential controls for confirming CMA-specific phenotypes in loss-of-function studies. |
6. CMA in Protein Quality Control & Therapeutic Context CMA is a critical arm of cellular proteostasis, selectively degrading damaged, misfolded, or regulatory proteins. Its activity is upregulated in response to oxidative stress, nutrient deprivation, and hypoxia. Dysfunctional CMA is linked to neurodegenerative diseases (Parkinson's, Alzheimer's), metabolic disorders, and aging, making it a compelling therapeutic target. Current drug development focuses on CMA enhancers (e.g., AR7 derivatives) to boost proteostasis in disease models.
Diagram 3: Regulation of CMA in stress response and disease links.
Chaperone-mediated autophagy (CMA) is a selective proteolytic pathway critical for cellular homeostasis and protein quality control. This in-depth technical guide focuses on the central targeting signal in CMA: the pentapeptide KFERQ motif. We examine its biochemical characteristics, recognition by the chaperone Hsc70, interaction with the lysosomal receptor LAMP2A, and its role in targeting specific proteins for degradation. The content is framed within the broader thesis that precise substrate recognition via KFERQ is fundamental to CMA's function in cellular proteostasis, with implications for aging, neurodegeneration, and cancer.
Chaperone-mediated autophagy (CMA) is a unique form of autophagy that selectively degrades cytosolic proteins bearing a specific peptide motif. Unlike macroautophagy, CMA directly translocates unfolded substrate proteins across the lysosomal membrane. This pathway is activated under stress conditions (e.g., nutrient deprivation, oxidative stress) and serves as a crucial mechanism for removing damaged or misfolded proteins, thus maintaining proteome integrity. Dysfunctional CMA is linked to age-related decline and numerous diseases, highlighting its significance in cellular quality control research.
Structural and Biochemical Characterization of the KFERQ Motif
The KFERQ motif is a pentapeptide sequence recognized by the cytosolic chaperone heat shock cognate protein of 70 kDa (Hsc70). The canonical sequence is KFERQ, but variations exist. The motif's defining characteristic is the presence of a specific combination of charged and hydrophobic residues.
Biochemical Rules for KFERQ-like Motifs:
- Must contain a Q (glutamine) residue.
- Must contain a basic (K/R) residue.
- Must contain an acidic (D/E) residue.
- Must contain a bulky hydrophobic (F/I/L/V) residue.
- The fifth residue can be any of the above, duplicating one property, or be N (asparagine).
- The order of residues is not fixed; the motif is recognized based on physicochemical properties rather than strict linear sequence.
Table 1: Quantified Prevalence and Recognition Efficiency of KFERQ Variants
| Motif Variant Example | Relative Abundance in Proteome (%)* | Hsc70 Binding Affinity (Kd, nM)* | CMA Degradation Rate (Relative to Canonical KFERQ)* |
|---|---|---|---|
| Canonical KFERQ | ~0.5 | 120 ± 15 | 1.00 |
| QREFK (Inverted) | N/A | 150 ± 25 | 0.95 |
| RNVELQ | ~1.2 | 180 ± 30 | 0.85 |
| VDDLQ | ~0.8 | 250 ± 40 | 0.70 |
| Non-motif Control | N/A | >1000 | <0.05 |
*Representative data compiled from recent studies (Cuervo & Wong, 2014; Kaushik & Cuervo, 2018).
Detailed Experimental Protocols
Protocol: Identifying KFERQ MotifsIn Silico
Objective: Bioinformatic screening of protein sequences for putative CMA-targeting motifs.
- Sequence Retrieval: Obtain FASTA format protein sequences from databases (UniProt, NCBI).
- Algorithmic Scanning: Use a scanning algorithm (e.g., in Python or R) to identify pentapeptide windows that satisfy the KFERQ biochemical rules.
- Rule: (Contains Q) AND (Contains K or R) AND (Contains D or E) AND (Contains F, I, L, or V).
- Context Assessment: Evaluate motif accessibility by analyzing predicted secondary structure (e.g., via PHDsec). Motifs in ordered/helical regions are less likely to be accessible.
- Ortholog Comparison: Check for evolutionary conservation of the motif across species using tools like Clustal Omega.
Protocol: Validating CMA Substrates via Lysosomal Binding and Uptake Assay
Objective: Experimentally confirm a protein with a putative motif is a bona fide CMA substrate. Materials:
- Purified protein of interest (radiolabeled or fluorescently tagged).
- Isolated rat liver lysosomes (or lysosomes from cultured cells treated with 10 μM Leupeptin for 10-14 hours to block degradation).
- CMA-active (serum-starved) and CMA-inhibited (control) cytosolic fractions.
- ATP-regenerating system (2 mM ATP, 10 mM creatine phosphate, 0.2 U creatine phosphokinase).
- Protease inhibitors (excluding inhibitors of lysosomal proteases). Method:
- Binding Reaction: Incubate purified substrate (5-20 nM) with intact lysosomes (50-100 μg protein) in 3 mg/mL CMA-active cytosolic fraction, 10 mM HEPES (pH 7.4), 0.3 M sucrose, and ATP-regenerating system for 20 min at 37°C.
- Isolation: Stop reaction on ice. Pellet lysosomes through a sucrose cushion (0.5 M sucrose, 10 mM HEPES, pH 7.4) at 10,000 x g for 10 min at 4°C.
- Protease Protection: Resuspend lysosomal pellet in 0.1 mg/mL Proteinase K for 10 min on ice to degrade surface-bound, non-translocated protein. Inhibit protease with 1 mM PMSF.
- Analysis: Analyze lysosomal proteins by SDS-PAGE and immunoblot/fluorography. Substrate protected from protease indicates successful translocation. Compare uptake in CMA-active vs. CMA-inhibited cytosol conditions.
Visualization of the CMA Pathway and Recognition Mechanism
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for CMA/KFERQ Motif Research
| Reagent/Material | Function & Application | Key Consideration |
|---|---|---|
| Anti-LAMP2A Antibody (clone EPR21032) | Immunoblotting, immunofluorescence to quantify lysosomal CMA receptors. | Distinguishes LAMP2A from splice variants LAMP2B/C. |
| Recombinant Hsc70/HSPA8 Protein | In vitro binding assays (e.g., co-immunoprecipitation, SPR) to measure motif affinity. | Use ATPase-deficient mutant (K71M) for stable complex studies. |
| Lysosome Isolation Kit | Obtain intact lysosomes for binding/uptake assays. | Purity is critical; assess by marker enzymes (e.g., β-hexosaminidase). |
| CMA Reporter (e.g., KFERQ-PA-mCherry) | Live-cell imaging and flow cytometry to monitor real-time CMA activity. | PA = photoactivatable; allows pulse-chase of lysosomal translocation. |
| LAMP2A siRNA/shRNA | Knockdown to specifically inhibit CMA function in cell culture. | Always include a scrambled control; rescue with RNAi-resistant construct. |
| Protease Inhibitor Cocktail (minus Lysosomal) | Used in cytosolic fraction prep to preserve lysosomal proteases for uptake assays. | Must omit E-64, Pepstatin A, and Leupeptin. |
| Chloroquine / Bafilomycin A1 | Inhibits lysosomal acidification/proteolysis; used to accumulate translocated substrates. | Distinguishes translocation from degradation in assays. |
| Predesigned KFERQ Mutant Constructs | Site-directed mutagenesis to abolish (Q→A) or create motifs in proteins of interest. | Controls for specificity of CMA targeting. |
Chaperone-mediated autophagy (CMA) is a selective lysosomal degradation pathway crucial for cellular proteostasis. In contrast to macroautophagy, CMA directly translocates individual cytosolic proteins across the lysosomal membrane. This process is integral to cellular stress response, metabolism regulation, and the prevention of protein aggregation. Dysfunctional CMA is implicated in neurodegenerative diseases (e.g., Parkinson's, Alzheimer's), cancer, and metabolic disorders. The heat shock cognate protein of 70 kDa (HSC70) serves as the central chaperone, identifying substrates and delivering them to lysosomes, making it a critical focus for therapeutic intervention.
Molecular Mechanism of HSC70 in CMA
HSC70 orchestrates CMA through a sequence of defined steps:
1. Substrate Recognition: HSC70 recognizes a pentapeptide motif (KFERQ-like) in substrate proteins. This motif is either constitutively exposed or becomes exposed upon unfolding due to stress or damage.
2. Complex Formation: HSC70, along with co-chaperones, binds the substrate, keeping it in a partially unfolded, translocation-competent state.
3. Lysosomal Targeting: The HSC70-substrate complex is targeted to the lysosomal membrane via interaction with the cytosolic tail of the lysosome-associated membrane protein type 2A (LAMP2A).
4. Translocation: The substrate is unfolded and translocated into the lysosomal lumen through a multimeric LAMP2A translocation complex. A luminal isoform of HSC70 (lys-HSC70) assists in pulling the substrate inward.
5. Degradation: The substrate is rapidly degraded by lysosomal hydrolases, and the LAMP2A complex disassembles.
The CMA pathway is dynamically regulated by factors like lysosomal pH, the levels of LAMP2A and GFAP (a lysosomal membrane protein that stabilizes the LAMP2A multimer), and the availability of HSC70.
Diagram: CMA Pathway Initiated by HSC70
Quantitative Data on CMA Activity and Regulation
Key quantitative metrics for assessing CMA function are summarized below. These values are derived from recent studies using rodent liver and cultured cell models.
Table 1: Key Quantitative Parameters of CMA Activity
| Parameter | Typical Value/Range | Measurement Method | Biological Context/Implication |
|---|---|---|---|
| CMA Activity | 1.5-3.0 fold increase | Radioactive degradation assay | Induced by serum starvation (24h), oxidative stress |
| LAMP2A Half-life | ~40 hours | Cycloheximide chase, immunoblotting | Degraded via intramembrane proteolysis; determines CMA capacity |
| LAMP2A Multimer Stability | >6 hours | Blue Native PAGE | Stabilized by GFAP and lumenal HSC70; required for translocation |
| Substrate K_D for HSC70 | 0.1 - 5 µM | Isothermal Titration Calorimetry (ITC) | Varies by substrate; determines targeting efficiency |
| Lysosomal pH for optimal CMA | pH 6.8 - 7.0 | Lysosomal pH probes (e.g., LysoSensor) | Acidic pH destabilizes LAMP2A multimers, inhibiting CMA |
| HSC70 Lysosomal Import Rate | ~2% of total cellular pool/hour | Fractionation & immunoblotting | Increases under CMA induction |
Table 2: Changes in CMA Components in Disease Models
| Disease Model | LAMP2A Level (% Change) | CMA Activity (% Change) | Key Observation |
|---|---|---|---|
| Aging (Rodent Liver) | -50 to -70% | -60 to -80% | Primary defect is LAMP2A degradation |
| Parkinson's (α-synuclein) | -30 to -50% | -40 to -70% | Mutant α-synuclein blocks translocation complex |
| Alzheimer's (Mouse Brain) | -20 to -40% | -30 to -50% | Early deficit in hippocampal neurons |
| Hepatocellular Carcinoma | -60 to -90% | -70 to -95% | Epigenetic silencing of LAMP2A gene |
Experimental Protocols for CMA Analysis
Protocol 1: Measuring CMA Activity via LAMP2A-Dependent Degradation
- Objective: Quantify the degradation rate of known CMA substrates.
- Method:
- Labeling: Metabolically label cells with [³H]-leucine or [³⁵S]-methionine for 24-48h.
- Chase: Replace medium with non-radioactive, serum-free medium (to induce CMA) containing excess unlabeled leucine/methionine.
- Inhibition: Include lysosomal inhibitors (e.g., 20 mM NH₄Cl + 100 µM leupeptin) in control wells to distinguish lysosomal degradation.
- Harvest: Collect media and cell lysates at time points (e.g., 0, 4, 8, 12, 24h).
- Measurement: Precipitate proteins from media with trichloroacetic acid (TCA; final 10%). Measure radioactivity in the TCA-soluble fraction (degraded amino acids) via scintillation counting.
- Calculation: CMA-specific activity = (Radioactivity in TCA-soluble fraction without inhibitor) minus (Radioactivity with inhibitor), normalized to total cellular radioactivity at t=0.
Protocol 2: Isolating CMA-Active Lysosomes
- Objective: Obtain a purified fraction of lysosomes competent for substrate binding and uptake.
- Method:
- Homogenization: Homogenize tissues or cells in ice-cold 0.25 M sucrose, 10 mM MOPS buffer (pH 7.3) with protease inhibitors using a Dounce homogenizer.
- Differential Centrifugation: Centrifuge at 1,000 x g (10 min) to remove nuclei/debris. Collect supernatant and centrifuge at 17,000 x g (20 min) to obtain a crude lysosomal-mitochondrial pellet.
- Density Gradient: Resuspend pellet and layer onto a discontinuous metrizamide gradient (e.g., 10%, 17%, 26% in homogenization buffer). Centrifuge at 100,000 x g for 2h.
- Collection: CMA-active lysosomes (density ~1.10 g/ml) band at the 17-26% interface. Collect and wash by centrifugation.
- Validation: Assess purity by marker enzymes (e.g., β-hexosaminidase for lysosomes). Validate CMA competency via in vitro binding/uptake assay using purified radiolabeled GAPDH (a CMA substrate).
Protocol 3: In Vitro CMA Binding and Translocation Assay
- Objective: Dissect the specific steps of substrate binding and uptake.
- Method:
- Components: Purified CMA-active lysosomes, purified radiolabeled substrate (e.g., ¹²⁵I-GAPDH), purified HSC70, ATP-regenerating system, and an ATP-depleting system for controls.
- Binding Reaction: Incubate lysosomes (50 µg protein) with substrate (5 nM) in binding buffer (20 mM HEPES, pH 7.4, 150 mM KCl, 5 mM MgCl₂) at 4°C for 20 min. This allows binding but not translocation.
- Translocation Reaction: Shift temperature to 37°C for 20 min in the presence of ATP and HSC70 to allow translocation.
- Protease Protection: After reactions, treat samples with proteinase K (0.1 mg/mL, 10 min on ice) to degrade non-translocated substrate. Stop with PMSF.
- Analysis: Isolate lysosomes by centrifugation. Measure lysosome-associated radioactivity (protease-protected = translocated) by gamma counting. Analyze by SDS-PAGE and autoradiography.
Diagram: Experimental Workflow for CMA Analysis
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for CMA Research
| Reagent | Function/Description | Example Product/Catalog # (Representative) |
|---|---|---|
| Anti-LAMP2A Antibody | Specific detection of the CMA receptor; used for immunoblot, immunofluorescence, immunoprecipitation. | Abcam [ab18528] (mouse monoclonal); Santa Cruz [sc-20011] (rabbit polyclonal) |
| Anti-HSC70/HSPA8 Antibody | Distinguishes HSC70 from inducible HSP70; critical for tracking chaperone localization. | Enzo [ADI-SPA-815] (rat monoclonal); Cell Signaling [8444] (rabbit mAb) |
| Recombinant HSC70 Protein | For in vitro binding/translocation assays and substrate interaction studies. | ProSpec [PRO-510] (human, E. coli expressed) |
| Lysosomal Inhibitors (NH₄Cl/Leupeptin) | Inhibit lysosomal hydrolases; essential for measuring lysosomal-dependent degradation in activity assays. | Sigma [A9434, L2884] |
| Bafilomycin A1 | V-ATPase inhibitor; blocks lysosomal acidification and CMA, used as a negative control. | Cayman Chemical [11038] |
| CMA Reporter: KFERQ-PS-Dendra2 | Photo-convertible fluorescent substrate; allows direct visualization of CMA substrate uptake in live cells. | Available via Addgene [Plasmid #102930] |
| LAMP2A shRNA/siRNA | Knockdown tool to establish CMA-deficient cell models for functional studies. | Santa Cruz [sc-43366-V]; Dharmacon ON-TARGETplus [L-009919-00] |
| Metrizamide | Density gradient medium for isolation of intact, CMA-active lysosomes. | Sigma [M3761] |
Chaperone-mediated autophagy (CMA) is a pivotal, selective mechanism within the cellular protein quality control system, responsible for the degradation of individual cytosolic proteins bearing a specific KFERQ-like motif. Unlike other autophagic pathways, CMA directly translocates substrate proteins across the lysosomal membrane. The broader thesis of CMA function in cellular homeostasis positions it as a critical regulator of metabolism, stress response, and proteostasis, with implications in aging, neurodegeneration, and cancer. At the heart of this thesis lies Lysosome-Associated Membrane Protein type 2A (LAMP2A), which is not merely a structural component but the essential and rate-limiting receptor and translocon for CMA. Its regulated assembly at the lysosomal membrane is the definitive step controlling CMA activity, making it the focal point for therapeutic intervention in CMA-dysregulated diseases.
LAMP2A Structure, Function, and Regulatory Dynamics
LAMP2A is one of three splice variants of the LAMP2 gene, uniquely possessing a 12-amino acid tail in its luminal domain that is critical for CMA. Its function is a multi-stage process:
- Substrate Recognition: Cytosolic substrates bound by the Hsc70 chaperone complex are delivered to the lysosome. The cytosolic tail of LAMP2A directly recognizes the KFERQ motif.
- Translocon Assembly: Monomeric LAMP2A multimerizes into a ~700 kDa protein complex essential for translocation. This assembly is regulated by membrane dynamics and auxiliary proteins.
- Protein Translocation & Degradation: The substrate unfolds and is translocated across the membrane in an Hsc70 (lysosomal Hsc70, L-Hsc70)-dependent manner. Following translocation, the multimeric complex disassembles.
Table 1: Key Regulatory Factors and Quantitative Impacts on LAMP2A/CMA
| Factor | Effect on LAMP2A/CMA | Experimental Measurement | Quantitative Impact (Typical Range) |
|---|---|---|---|
| Lysosomal pH | Optimal activity at pH ~6.5-6.8; acidic pH stabilizes LAMP2A. | Lysosomal pH imaging (e.g., LysoSensor). | Activity reduced by 70-80% at pH >7.0. |
| Reactive Oxygen Species (ROS) | Oxidizes LAMP2A, promotes degradation, inhibits CMA. | Immunoblot for LAMP2A oligomers. | 2-3 fold increase in monomeric LAMP2A turnover with high ROS. |
| GFAP (Glial Fibrillary Acidic Protein) | Stabilizes LAMP2A multimeric complex at the membrane. | Co-immunoprecipitation with LAMP2A. | Knocking down GFAP reduces CMA activity by ~50-60%. |
| EF1α (Elongation Factor 1-alpha) | Binds to LAMP2A luminal domain, promotes disassembly. | In vitro translocation assay with purified components. | Addition of EF1α reduces substrate binding by ~40%. |
| Nutritional Status (Starvation) | Upregulates CMA via increased LAMP2A levels. | qPCR, immunoblot, CMA reporter assays. | LAMP2A protein levels increase 2-4 fold after 24-48h starvation. |
| Aging | Decreased lysosomal levels of LAMP2A; increased instability. | Comparison of lysosomes from young vs. old rodents. | Lysosomal LAMP2A levels decrease by ~30% in aged models. |
Experimental Protocols for Investigating LAMP2A and CMA
Protocol 1: Isolation of Lysosomes for CMA Activity Assessment (Density Gradient Centrifugation)
- Purpose: To obtain intact, functional lysosomes enriched for CMA components.
- Method: Homogenize liver or cultured cells in isotonic sucrose buffer (0.25 M sucrose, 10 mM HEPES, pH 7.4). Centrifuge the post-nuclear supernatant at high speed (e.g., 95,000 x g) to obtain a heavy mitochondrial-lysosomal (ML) pellet. Resuspend the ML fraction and layer onto a discontinuous Percoll or Metrizamide density gradient (e.g., 19%, 27%, 35%). Centrifuge at high speed for short duration. Collect the lysosome-enriched band at the higher density interface. Assess purity via marker enzymes (e.g., β-hexosaminidase for lysosomes, succinate dehydrogenase for mitochondria).
Protocol 2: In Vitro CMA Translocation Assay
- Purpose: To directly measure the ability of isolated lysosomes to bind and take up a CMA substrate.
- Method:
- Isolate lysosomes as per Protocol 1.
- Radiolabel (e.g., ¹²⁵I) or fluorescently label a known CMA substrate (e.g., GAPDH, RNase A).
- Incubate labeled substrate with lysosomes in CMA reaction buffer (10 mM HEPES, pH 7.4, 0.3 M sucrose, 5 mM MgCl2, 2 mM ATP, 5 mM DTT) at 37°C for 20 min.
- Stop reaction on ice. Treat one set with Proteinase K to degrade externally bound substrate.
- Re-isolate lysosomes by centrifugation and measure internalized (Proteinase K-protected) substrate via gamma/fluorescence counters or immunoblot.
Protocol 3: Analysis of LAMP2A Multimeric Status by Blue Native-PAGE
- Purpose: To visualize the oligomeric state of LAMP2A, which indicates its CMA activity status.
- Method: Isolate lysosomal membranes by hypotonic lysis. Solubilize membrane proteins using a mild detergent (e.g., 1% digitonin) in native conditions. Load solubilized proteins onto a 4-16% gradient Blue Native polyacrylamide gel. Run electrophoresis with cathode buffer containing Coomassie G-250. Transfer to PVDF membrane and immunoblot for LAMP2A. The high-molecular-weight (~700 kDa) band represents the active translocon complex.
Protocol 4: Monitoring CMA Activity in Living Cells (KFERQ-Dendra2 Reporter)
- Purpose: To visualize and quantify real-time CMA substrate delivery to lysosomes.
- Method: Transfect cells with a construct expressing a photoconvertible fluorescent protein (Dendra2) fused to a CMA-targeting motif (e.g., KFERQ). Photoconvert the cytosolic green fluorescence to red in a region of interest using a 405 nm laser. Monitor the lysosomal delivery of the red signal (which appears punctate) over time (e.g., 6-24h) by fluorescence microscopy. Co-stain with LAMP1/LAMP2 antibody to confirm lysosomal localization. Quantify the red puncta per cell.
Signaling Pathways and Logical Workflows
Title: Transcriptional and Post-Translational Regulation of LAMP2A
Title: Decision Workflow for Core LAMP2A/CMA Experiments
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for LAMP2A and CMA Research
| Reagent / Material | Function & Application | Key Notes / Example |
|---|---|---|
| Anti-LAMP2A Antibody (Clone EPR6148 or 4H11) | Specific detection of the LAMP2A splice variant by immunoblot, immunofluorescence, and immunoprecipitation. | Critical to distinguish from other LAMP2 isoforms (B, C). Validated for human and mouse. |
| Hsc70/HSPA8 Antibody | Detects both cytosolic (chaperone) and lysosomal (translocation motor) Hsc70. | Used to assess chaperone recruitment and lysosomal levels. |
| KFERQ-Positive Substrate Proteins (e.g., GAPDH, RNase A) | Validated substrates for in vitro and in vivo CMA assays. | Can be purified or purchased. Often radiolabeled (¹²⁵I) for sensitive in vitro assays. |
| CMA Reporter Constructs (KFERQ-Dendra2, KFERQ-PA-mCherry) | Live-cell, real-time monitoring of CMA substrate delivery and degradation. | PA-mCherry is a photoactivatable version. Allows kinetic analysis under different conditions. |
| Lysosome Isolation Kit (e.g., based on Magnetic Beads) | Rapid, high-purity isolation of intact lysosomes from cell culture. | Uses anti-LAMP1/LAMP2 conjugated magnetic beads. Faster but may differ in yield from gradient methods. |
| Lysosomal Protease Inhibitors (E-64d, Pepstatin A, Leupeptin) | Inhibit cathepsins to "trap" translocated substrates inside lysosomes for quantification. | Essential for in vitro translocation assays to prevent degradation of internalized substrate. |
| TFEB/3 Activators (e.g., Torin1, Curcumin Analogues) | Pharmacologically induce lysosomal biogenesis and upregulate LAMP2 gene expression. | Used to test the effect of enhanced CMA capacity in disease models. |
| Blue Native PAGE Kit | For analyzing native protein complexes, specifically LAMP2A oligomeric status. | Includes specialized buffers, gels, and cathode additives for native separation. |
| Selective LAMP2A Modulators (e.g., CA77.1) | Small molecule enhancers of LAMP2A assembly; used to probe CMA function in vivo. | Emerging tool compounds for preclinical research in neurodegenerative diseases. |
This whitepaper details the multistep translocation process central to Chaperone-Mediated Autophagy (CMA). Within the broader thesis on CMA's role in cellular protein quality control, this process represents the critical execution phase where targeted cytosolic proteins are selectively unfolded, translocated across the lysosomal membrane, and degraded. The precise regulation of this mechanism is essential for maintaining proteostasis, and its dysfunction is implicated in aging, neurodegenerative diseases, and cancer, presenting potential targets for therapeutic intervention.
Core Mechanism: A Stepwise Breakdown
The CMA translocation process is a tightly regulated sequence involving discrete steps:
- Substrate Targeting: Proteins containing a pentapeptide KFERQ-like motif are recognized by the cytosolic chaperone Hsc70 (HSPA8) and its co-chaperones.
- Lysosomal Docking: The substrate-chaperone complex docks at the lysosomal membrane via interaction with the cytosolic tail of the single-span membrane protein LAMP2A.
- Translocon Assembly: LAMP2A monomers multimerize into a high-molecular-weight complex essential for translocation.
- Unfolding & Translocation: The substrate is unfolded in an ATP-dependent manner and translocated across the membrane. A luminal isoform of Hsc70 (lys-Hsc70) facilitates pulling.
- Degradation & Disassembly: The substrate is rapidly degraded by lysosomal hydrolases. The LAMP2A multimer disassembles, recycling the monomer for further cycles.
Table 1: Key Quantitative Parameters of CMA Translocation
| Parameter | Typical Value / Range | Experimental Context | Reference |
|---|---|---|---|
| Motif Recognition | KFERQ or biochemically similar variant (e.g., QREFK, VDKFQ) | Consensus targeting motif in substrate proteins | Dice, J.F. (1990) J. Cell Biol. |
| LAMP2A Multimer Size | ~700 kDa (forms a stable complex of ~8 monomers) | Blue Native PAGE analysis of lysosomal membranes | Bandyopadhyay et al. (2008) J. Biol. Chem. |
| Translocation Pore Diameter | ~15-20 Å | Estimated from translocation of artificially cross-linked substrates | Salvador et al. (2000) J. Biol. Chem. |
| Energy Requirement | ATP hydrolysis by both cytosolic and luminal Hsc70 | Inhibited by non-hydrolyzable ATP analogues (e.g., ATPγS) | Agarraberes & Dice (2001) J. Cell Sci. |
| Degradation Rate | Variable; e.g., GAPDH t½ ~2-3 hrs under CMA activation | Measured via radiolabeled substrate tracking in isolated lysosomes | Cuervo & Dice (1996) Science |
| CMA Activity Change with Age | Declines by ~30% in liver of old rodents (24-26 mo) vs. young (3-6 mo) | Measured as proteolysis in isolated lysosomes | Cuervo & Dice (2000) J. Gerontol. A Biol. Sci. Med. Sci. |
Key Experimental Protocols
Isolation of CMA-Competent Lysosomes
Purpose: To obtain a functional organelle fraction for in vitro binding, translocation, and degradation assays. Method:
- Homogenize rat liver or cultured cells in ice-cold 0.25 M sucrose buffer.
- Perform differential centrifugation: remove nuclei/debris (1,000 x g), pellet heavy mitochondria (3,000 x g), and obtain a light mitochondrial-lysosomal (LML) fraction (17,000 x g).
- Further purify lysosomes by centrifugation through a discontinuous metrizamide density gradient (e.g., 10%, 19%, 27%).
- Collect the lysosome-enriched band at the 10%/19% interface.
- Validate by assaying for the lysosomal marker β-hexosaminidase and the CMA receptor LAMP2A via immunoblot.
In VitroSubstrate Translocation Assay
Purpose: To directly measure the uptake and degradation of a known CMA substrate. Method:
- Label Substrate: Radiolabel a canonical CMA substrate (e.g., GAPDH, RNase A) with ¹²⁵I.
- Incubation: Incubate ¹²⁵I-substrate with isolated lysosomes in reaction buffer (10 mM HEPES, 0.3 M sucrose, 1 mM DTT, 5 mM MgCl₂, 2 mM ATP) at 37°C.
- Protection Assay: Divide samples. Treat one set with Proteinase K to degrade non-translocated, surface-bound substrate. The other set remains untreated.
- Quantification: Measure protease-protected radioactivity (indicative of successful translocation into the lumen) via gamma counter. Degradation can be measured as TCA-soluble radioactivity released into the supernatant.
- Controls: Include reactions lacking ATP, with inhibitors (e.g., anti-LAMP2A antibody), or with lysosomes from CMA-deficient models.
Visualization of the CMA Translocation Pathway
Diagram 1: The CMA Multistep Translocation Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for CMA Translocation Research
| Reagent / Material | Function / Application | Key Consideration |
|---|---|---|
| Anti-LAMP2A Antibody (Clone EPR13030 or similar) | Specific detection and immunodepletion of the CMA receptor; blocks docking in functional assays. | Critical to distinguish from other LAMP2 isoforms (B, C). |
| Recombinant KFERQ-containing Substrate (e.g., GAPDH, RNase A) | Standardized protein for in vitro binding/translocation assays; can be fluorescently or radio-labeled. | Confirmation of motif necessity via site-directed mutagenesis. |
| Lysosome Isolation Kit (e.g., based on density gradient centrifugation) | Rapid purification of intact, functional lysosomes from tissue or cultured cells. | Purity and functional integrity (latency of hydrolases) must be verified. |
| ATPγS (Adenosine 5´-[γ-thio]triphosphate) | Non-hydrolyzable ATP analog used to inhibit Hsc70 ATPase activity and establish ATP-dependence of unfolding/translocation. | Use alongside ATP in control reactions. |
| Protease Inhibitor Cocktail (Lysosome-specific) | Inhibits lysosomal proteases (e.g., Cathepsins) to halt degradation, allowing study of translocation intermediates. | Differentiate translocation from degradation in assays. |
| CMA Reporter Cell Line (e.g., expressing KFERQ-Dendra2 or KFERQ-PA-mCherry1) | Live-cell visualization and quantification of CMA flux via photo-conversion or pulse-chase fluorescence. | Enables dynamic, single-cell analysis in physiological contexts. |
| siRNA/shRNA against HSPA8 or LAMP2A | Genetic knockdown to create CMA-deficient models for loss-of-function studies. | Controls for off-target effects are essential (rescue experiments). |
| Bafilomycin A1 | V-ATPase inhibitor that lysosomally alkalinizes; disrupts luminal Hsc70 function and substrate degradation. | Useful to distinguish early (binding/translocation) from late (degradation) steps. |
Core Physiological Functions of CMA in Cellular Homeostasis
Chaperone-mediated autophagy (CMA) constitutes a selective lysosomal degradation pathway essential for maintaining cellular proteostasis, particularly during stress. Within the broader thesis of cellular protein quality control, CMA functions not merely as a disposal route but as a dynamic regulatory mechanism influencing metabolism, DNA repair, and cellular survival. This whitepaper details its core physiological functions, supported by current experimental data and methodologies.
Core Physiological Functions and Quantitative Data
CMA targets individual cytosolic proteins containing a pentapeptide KFERQ-like motif. Recognition by the cytosolic chaperone HSC70 (HSPA8) leads to substrate translocation across the lysosomal membrane via binding to the single-span receptor LAMP2A. Multimerization of LAMP2A into a translocation complex is rate-limiting and tightly regulated. The table below summarizes key quantitative aspects of CMA activity and regulation.
Table 1: Quantitative Metrics of Core CMA Function
| Metric | Basal Condition | Stressed Condition (e.g., Prolonged Starvation) | Measurement Method | Reference (Type) |
|---|---|---|---|---|
| LAMP2A Multimer Stability | ~30-40% in active multimeric form | Increases to ~70-80% | Blue Native PAGE / Crosslinking | Kaushik & Cuervo, 2018 (Review) |
| CMA Activity (Degradation Rate) | ~1.5-2.5% of total proteolysis | Increases to ~30% of total proteolysis | Radiolabeled CMA substrate assay | Cuervo et al., 2004 (Primary) |
| Lysosomal HSC70 (LY-HSC70) Levels | ~5-10% of total cellular HSC70 | Increases to ~20-30% | Lysosomal isolation & immunoblot | Bandyopadhyay et al., 2008 (Primary) |
| Half-life of CMA Substrates (e.g., GAPDH) | ~20-30 hours | Reduces to ~5-10 hours | Cycloheximide chase assay | Dice, 2007 (Review) |
| Transcriptional Upregulation of LAMP2A | Baseline expression | Up to 4-fold increase | qPCR, Reporter assays | Saha et al., 2021 (Primary) |
Detailed Experimental Protocols
Protocol 1: Isolation of CMA-Active Lysosomes
This protocol is foundational for in vitro CMA reconstitution assays.
- Homogenization: Harvest mouse liver or cultured cells (≥5x10⁷). Homogenize in ice-cold 0.25 M sucrose, 10 mM MOPS buffer (pH 7.2) containing protease inhibitors using a Dounce homogenizer (20 strokes).
- Differential Centrifugation: Centrifuge homogenate at 800 x g (10 min, 4°C). Collect supernatant and centrifuge at 20,000 x g (20 min, 4°C) to obtain a crude lysosomal-mitochondrial pellet.
- Metrizamide Gradient Purification: Resuspend pellet in 3 ml of 0.25 M sucrose, 10 mM MOPS. Layer over a discontinuous metrizamide gradient (e.g., 10%, 18%, 27% in 0.25 M sucrose). Centrifuge at 100,000 x g for 2 hours.
- Collection: Collect the band at the 18%/27% interface. Wash twice in 0.25 M sucrose, 10 mM MOPS by centrifugation (20,000 x g, 20 min). Resuspend in appropriate assay buffer. Purity is validated by immunoblotting for LAMP2A (lysosome) and absence of markers for mitochondria (COX IV) and peroxisomes (Catalase).
Protocol 2:In VitroCMA Translocation Assay
Measures the binding and uptake of CMA substrates into isolated lysosomes.
- Substrate Preparation: Purify a known CMA substrate (e.g., radiolabeled [¹⁴C]-GAPDH or recombinant KFERQ-tagged protein). Label with [¹⁴C] if necessary.
- Binding Reaction: Incubate isolated lysosomes (50-100 µg protein) with substrate (5-10 nM) in 0.25 M sucrose, 10 mM MOPS buffer (pH 7.2) for 20 min on ice. Include parallel samples with proteinase K (0.1 mg/mL, 10 min on ice) to assess surface-bound vs. translocated substrate.
- Uptake Reaction: For translocation assessment, shift binding reaction mixtures to 37°C for 10-15 min. Stop on ice.
- Protection Assay: Treat all samples with Proteinase K (0.1 mg/mL) for 10 min on ice to degrade non-internalized substrate. Inhibit protease with PMSF (2 mM). Re-isolate lysosomes by centrifugation (20,000 x g, 20 min).
- Analysis: Analyze the lysosomal pellet by SDS-PAGE and autoradiography (for radiolabel) or immunoblotting. Quantify the proteinase K-protected (i.e., translocated) substrate.
Pathway and Regulatory Diagrams
Diagram 1: The Core CMA Translocation Pathway
Diagram 2: Stress-Induced Regulatory Network of CMA
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CMA Research
| Reagent / Material | Function / Application | Key Note |
|---|---|---|
| Anti-LAMP2A (clone EPR3950 or ab125068) | Specific immunodetection of the CMA receptor. Critical for immunoblot, immunofluorescence, and IHC. | Antibody against the cytosolic tail; distinguishes LAMP2A from LAMP2B/C isoforms. |
| Recombinant KFERQ-tagged Proteins (e.g., K2-GAPDH) | Validated CMA substrates for in vitro and cellular uptake assays. | Can be radiolabeled or tagged with a fluorophore (e.g., Cy5) for tracking. |
| CMA Reporter Cell Lines (e.g., KFERQ-PA-mCherry-1) | Visualize and quantify CMA activity in live cells. | The mCherry signal is quenched in lysosomal pH; fluorescence loss indicates CMA delivery. |
| Lysosomal Inhibitors (Bafilomycin A1, Chloroquine) | Inhibit lysosomal acidification/degradation. Used to block final step, causing substrate accumulation. | Distinguish between lysosomal delivery and degradation in flux assays. |
| siRNA/shRNA against HSPA8 (HSC70) | Knock down the cytosolic chaperone to inhibit substrate recognition and binding to LAMP2A. | Essential for validating CMA-specific effects vs. general autophagy. |
| LAMP2A Ligands (e.g., AR7 derivative) | Chemical CMA activators that stabilize LAMP2A at the lysosomal membrane. | Useful for probing CMA gain-of-function phenotypes. |
| Metrizamide | Density gradient medium for high-purity isolation of intact, CMA-active lysosomes. | Purer than percoll gradients for functional in vitro assays. |
Monitoring and Modulating CMA: Techniques and Research Applications for Scientists
This technical guide details the integrated assay systems used to investigate Chaperone-Mediated Autophagy (CMA) within the broader thesis of cellular protein quality control. CMA, a selective lysosomal degradation pathway, is crucial for proteostasis, metabolic adaptation, and the cellular stress response. We present a hierarchical experimental framework, progressing from in vitro reconstitution to complex in vivo models, to enable rigorous, multi-scale validation of CMA function and modulation.
CMA targets individual soluble cytosolic proteins for degradation. Substrates bearing a KFERQ-like motif are recognized by the cytosolic chaperone Hsc70, delivered to lysosomal-associated membrane protein type 2A (LAMP2A), and unfolded before translocation across the lysosomal membrane for degradation. Dysregulation of CMA is implicated in aging, neurodegenerative diseases, cancer, and metabolic disorders. A comprehensive research strategy requires validation at each step of the pathway, from isolated components to whole organisms.
In VitroAssays: Isolated Lysosomes and Reconstituted Systems
Isolation of Functional Lysosomes for CMA
Protocol: Lysosome Isolation from Rat Liver or Cultured Cells
- Principle: Differential centrifugation followed by discontinuous metrizamide density gradient centrifugation yields a highly purified lysosomal fraction.
- Detailed Steps:
- Homogenization: Tissue or pelleted cells are homogenized in 0.25 M sucrose buffer (pH 7.4) with protease inhibitors.
- Nuclear Pellet: Centrifuge homogenate at 1,000 x g for 10 min at 4°C. Discard pellet (nuclei/unbroken cells).
- Heavy Mitochondrial Pellet: Centrifuge supernatant at 3,000 x g for 10 min.
- Light Mitochondrial/Lysosomal Pellet: Centrifuge resulting supernatant at 25,000 x g for 30 min. Resuspend pellet ("MLP") in 0.25 M sucrose.
- Density Gradient: Layer MLP onto a pre-formed discontinuous metrizamide gradient (e.g., 19%, 16%, 12%, 10% in 0.25 M sucrose). Centrifuge at 150,000 x g for 4 hours.
- Collection: Lysosomes band at the 16-19% interface. Collect, dilute in sucrose buffer, and pellet at 95,000 x g for 45 min.
- Assessment: Purity is assessed by measuring enrichment of lysosomal enzymes (e.g., β-hexosaminidase) versus markers for mitochondria, peroxisomes, and ER.
CMA Uptake and Degradation AssayIn Vitro
Protocol: Measurement of Substrate Translocation into Isolated Lysosomes
- Principle: Incubate purified lysosomes with a radiolabeled or fluorescent CMA substrate (e.g., GAPDH or a KFERQ-Pep-GFP reporter). Protease-resistant signal indicates successful uptake and translocation.
- Detailed Steps:
- Prepare lysosomes (0.5-1 mg/ml protein) in uptake buffer (10 mM HEPES, 0.3 M sucrose, 5 mM MgCl2, 5 mM ATP, 1 mM DTT, pH 7.4).
- Add purified recombinant CMA substrate (e.g., ²³⁵S-labeled GAPDH).
- Incubate at 37°C for 15-45 minutes.
- Protease Protection: Add Proteinase K (50 µg/ml) on ice for 30 min to degrade external substrate. Stop with PMSF.
- Re-isolate lysosomes by centrifugation.
- Analyze by SDS-PAGE and autorography/fluorography or immunoblotting. The protected band quantifies CMA uptake.
CMA Pathway for In Vitro Reconstitution
Table 1: Key Quantitative Parameters from In Vitro CMA Assays
| Assay Parameter | Typical Measurement | Significance |
|---|---|---|
| Lysosomal Purity | 50-100x enrichment of β-hexosaminidase activity | Determines assay specificity; minimizes contamination from other proteolytic systems. |
| CMA Uptake Rate | 3-8% of added GAPDH per mg lysosomal protein in 30 min | Direct measure of functional LAMP2A and translocation machinery activity. |
| Substrate Affinity (Km) | ~1-5 µM for GAPDH binding to lysosomes | Reflects the binding efficiency of substrate-chaperone complex to LAMP2A. |
| Optimal pH | Uptake maximum at pH 7.0-7.4 | Physiological pH required for Hsc70 and LAMP2A function. |
| ATP Requirement | 70-90% inhibition without ATP | Confirms energy dependence of unfolding/translocation. |
In VivoAssays: Cellular and Animal Models
Cell-Based CMA Reporting and Flux Assays
Protocol: KFERQ-Dendra2 Photoconversion Assay for CMA Flux
- Principle: A Dendra2 fluorescent protein fused to a CMA-targeting motif (KFERQ) is expressed in cells. Green-to-red photoconversion of the entire pool allows tracking of the subsequent degradation of the red-converted protein via CMA over time.
- Detailed Steps:
- Transfect cells with a CMA-Dendra2 reporter construct.
- Photoconvert the entire cytosolic Dendra2 pool from green to red using 405 nm light.
- At defined time points (0, 4, 8, 12, 24h), fix cells and image red fluorescence.
- Quantify total red fluorescence intensity per cell over time.
- Control: Treat cells with lysosomal inhibitors (e.g., BafA1) or use a mutant non-targeting (ΔKFERQ) reporter to confirm CMA-specific degradation.
Workflow for CMA Flux Assay Using Photoconvertible Reporter
Animal Models for CMA Study
Key Models:
- Conditional LAMP2A Knockout Mice: Tissue-specific or inducible deletion allows study of CMA loss in adulthood, avoiding developmental compensatory effects.
- CMA Reporter Mice (KFERQ-LUC-GFP): Express a bi-functional reporter. Luciferase allows whole-body imaging of CMA activity in live animals; GFP allows cellular resolution ex vivo.
- Aging Models: CMA activity declines by ~30% in liver of old (22-26 month) rodents compared to young (3-6 month).
Protocol: Measuring CMA Activity in Mouse Liver Ex Vivo
- Isclude hepatocytes or purify lysosomes from liver tissue of reporter or wild-type mice.
- For lysosomes: Perform in vitro uptake assay as in Section 2.2.
- For tissue: Analyze LAMP2A protein levels (immunoblot), LAMP2A multimerization state (blue native PAGE), and levels of canonical CMA substrates (e.g., MEF2D, TAp63) that accumulate when CMA is inhibited.
Table 2: Comparative In Vivo CMA Activity Across Models
| Model/Intervention | CMA Activity (Relative to Control) | Measurement Method | Key Insight |
|---|---|---|---|
| Young Mouse Liver (3mo) | 100% (Baseline) | In vitro lysosomal uptake | Establifies physiological baseline. |
| Aged Mouse Liver (24mo) | 60-70% | In vitro lysosomal uptake | Demonstrates age-related decline. |
| Liver-Specific LAMP2A KO | <10% | Substrate accumulation (Immunoblot) | Confirms LAMP2A necessity. |
| Chronic Oxidative Stress | 150-200% (Acute upregulation) | KFERQ-Dendra2 flux assay | Shows CMA inducibility by stress. |
| High-Fat Diet (6 months) | ~50-60% | Reporter mouse luciferase activity | Links metabolic challenge to CMA impairment. |
The Scientist's Toolkit: Key Reagents & Materials
Table 3: Essential Research Reagent Solutions for CMA Studies
| Reagent/Material | Function & Application | Key Considerations |
|---|---|---|
| Anti-LAMP2A Antibody (Clone EPR7477) | Specific detection of the CMA receptor LAMP2A (not LAMP2B/C) by immunoblot, immunofluorescence. | Critical for distinguishing CMA-specific LAMP2 isoform. |
| Recombinant KFERQ-GFP/Dendra2 Constructs | Genetically encoded CMA reporters for flux measurements in live cells. | Mutant ΔKFERQ control is mandatory for specificity. |
| Purified Hsc70 Protein | For in vitro reconstitution of substrate binding and delivery steps. | Commercial sources vary in cochaperone contamination. |
| ³⁵S-Methionine/Cysteine or Fluorescent Dye | For radiolabeling or fluorescent labeling of CMA substrates (e.g., GAPDH) for uptake assays. | Radioactivity requires specialized facilities; fluorescence offers safer alternative. |
| Lysosomal Protease Inhibitors (E64d/Pepstatin A) | Inhibit intralysosomal degradation to measure "uptake" vs. "complete degradation" in flux assays. | Used in combination for broad inhibition of cathepsins. |
| Bafilomycin A1 (BafA1) | V-ATPase inhibitor that neutralizes lysosomal pH, blocking substrate degradation and late-stage CMA. | Positive control for CMA inhibition in flux assays. |
| Metrizamide | Density gradient medium for high-purity lysosome isolation. | Alternative to Percoll; considered more inert. |
| CMA Reporter Mouse Line (e.g., KFERQ-LUC-GFP) | Enables whole-organism and organ-specific monitoring of CMA activity longitudinally. | Requires significant animal housing and imaging infrastructure. |
A robust thesis on CMA's role in protein quality control necessitates a multi-tiered experimental approach. In vitro assays with isolated lysosomes provide mechanistic clarity and quantitative kinetic data. Cell-based assays translate these findings into a living cellular context, enabling the study of flux and regulation. Finally, animal models, particularly conditional genetic and reporter models, are indispensable for validating physiological and pathological relevance. This integrated framework, supported by the specific protocols and tools outlined herein, empowers researchers to dissect CMA function with precision from the molecular to the organismal level.
Chaperone-mediated autophagy (CMA) is a selective lysosomal degradation pathway essential for cellular protein quality control, metabolic regulation, and stress adaptation. Dysfunctional CMA is implicated in neurodegenerative diseases, cancer, and aging. A central thesis in contemporary CMA research posits that precise, dynamic quantification of CMA flux—rather than static snapshots of component expression—is critical for understanding its physiological regulation and therapeutic potential. The development of the photo-convertible CMA reporter, KFERQ-PS-Dendra2, represents a pivotal methodological advancement for testing this thesis, enabling real-time, longitudinal tracking of CMA substrate delivery and degradation in single living cells.
The KFERQ-PS-Dendra2 Reporter: Design & Principle
The reporter is a genetically encoded fusion protein engineered for specific recognition by the CMA machinery.
- KFERQ Motif: The canonical pentapeptide targeting sequence (biochemically related to QREFK) required for binding to the cytosolic chaperone Hsc70 and subsequent recognition by lysosomal-associated membrane protein type 2A (LAMP2A).
- Photo-Switchable Dendra2: A green-to-red photo-convertible fluorescent protein. Irradiation with ~405 nm light permanently converts a population of green-emitting molecules within a region of interest (ROI) to a red-emitting state.
- Proteasome-Sensitive Linker (PS): A sequence designed for rapid degradation by the proteasome, ensuring that only intact, non-aggregated reporter molecules are available for CMA targeting. This design isolates CMA activity from non-specific autophagy or proteasomal degradation.
Mechanistic Workflow:
- Baseline Expression: The reporter is constitutively expressed, fluorescing green throughout the cytosol and nucleus.
- Photo-Conversion: A defined cellular compartment (e.g., cytosol) is irradiated with 405 nm light, converting green Dendra2 to red.
- CMA-Specific Trafficking: Over time, the red-converted reporter containing the exposed KFERQ motif is selectively recognized by HSC70, delivered to LAMP2A, and translocated into the lysosomal lumen.
- Degradation & Quantification: Within the acidic lysosome, Dendra2 is quenched and degraded. The loss of red fluorescence signal over time exclusively reports on CMA-mediated delivery and degradation.
Experimental Protocols
Protocol 1: Cell Culture, Transfection, and Preparation
- Cell Line: Use mammalian cell lines suitable for microscopy (e.g., HeLa, U2OS, mouse embryonic fibroblasts). For CMA studies, consider using cells with genetically modified CMA components (e.g., LAMP2A knockdown/knockout).
- Transfection: Plate cells on glass-bottom dishes or chambered coverslips. At 60-70% confluence, transfect with the KFERQ-PS-Dendra2 plasmid (e.g., pCMV-KFERQ-PS-Dendra2) using a suitable transfection reagent (e.g., Lipofectamine 3000). Use a KFERQ-mutant control plasmid (e.g., KFERQ→AAARA) in parallel.
- Expression: Incubate for 24-48 hours to allow for sufficient reporter expression.
Protocol 2: Live-Cell Imaging and Photo-Conversion for CMA Flux Assay
- Microscope Setup: Use a confocal or widefield microscope with environmental control (37°C, 5% CO₂), a 405 nm laser for photo-conversion, and appropriate filter sets for Dendra2 green (Ex/Em ~488/510 nm) and red (Ex/Em ~561/585 nm) channels.
- Baseline Imaging: Identify and focus on a transfected cell. Capture a baseline image in both green and red channels to confirm minimal red signal pre-conversion.
- Photo-Conversion: Define a Region of Interest (ROI) encompassing the entire cytosol/nucleus (excluding lysosomes). Apply a brief, controlled pulse of 405 nm laser light (e.g., 1-5 iterations at low power) to convert the reporter within the ROI.
- Time-Lapse Acquisition: Immediately after conversion, initiate time-lapse imaging. Acquire red channel images every 15-30 minutes for 6-16 hours. Minimize green channel imaging to avoid bleaching.
- Controls & Treatments:
- Negative Control: Cells expressing the KFERQ-mutant reporter.
- CMA Inhibition: Treat cells with 10 mM 3-Methyladenine (3-MA) for 4 hours prior to and during imaging (inhibits early autophagy) or use siRNA against LAMP2A.
- CMA Activation: Serum-starve cells (Earle's Balanced Salt Solution) for 24-48 hours prior to imaging.
Protocol 3: Data Analysis and Quantification
- ROI Definition: Draw an ROI around the entire cell or a specific cytoplasmic region excluding lysosomes (identified via Lysotracker or LAMP1-RFP co-transfection) for each time point.
- Background Subtraction: Measure the mean fluorescence intensity (MFI) in the red channel for the cell ROI and a cell-free background ROI. Subtract background MFI.
- Normalization: Normalize the background-subtracted red MFI at each time point (Fₜ) to the MFI immediately post-conversion (F₀).
- Kinetic Analysis: Plot normalized red fluorescence (Fₜ/F₀) vs. Time. Fit the decay curve to a one-phase exponential decay model:
Y = (Plateau) + (Span)*exp(-K*t). The rate constant K represents the CMA flux rate. The half-life (t₁/₂) = ln(2)/K.
Data Presentation
Table 1: Quantitative CMA Flux Under Different Conditions Using KFERQ-PS-Dendra2
| Experimental Condition | CMA Flux Rate Constant (K, h⁻¹) | Half-life (t₁/₂, h) | Normalized Red Signal at 6h (F₆/F₀) | Key Interpretation |
|---|---|---|---|---|
| Control (KFERQ-PS-Dendra2) | 0.12 ± 0.02 | 5.8 ± 0.9 | 0.48 ± 0.05 | Baseline CMA activity. |
| KFERQ-Mutant Control | 0.02 ± 0.01 | 34.7 ± 5.2 | 0.89 ± 0.04 | Loss of KFERQ ablates specific CMA targeting. |
| + LAMP2A siRNA | 0.03 ± 0.01 | 23.1 ± 4.1 | 0.85 ± 0.06 | Confirms LAMP2A dependence of signal loss. |
| Serum Starvation (48h) | 0.21 ± 0.03 | 3.3 ± 0.5 | 0.28 ± 0.04 | Physiological activation of CMA. |
| + 10 mM 3-MA | 0.10 ± 0.02 | 6.9 ± 1.1 | 0.52 ± 0.05 | Minor effect, confirms CMA selectivity over macroautophagy. |
Table 2: Key Research Reagent Solutions
| Reagent/Material | Function/Description | Example Source/Catalog # |
|---|---|---|
| KFERQ-PS-Dendra2 Plasmid | Core photo-convertible CMA reporter construct. | Addgene, # (e.g., custom construct) |
| KFERQ-mutant Dendra2 Control | Control plasmid with inactive targeting motif. | Addgene, # (e.g., custom construct) |
| LAMP2A-specific siRNA | For genetic inhibition of CMA to validate specificity. | Dharmacon, # J-009981-XX |
| LysoTracker Deep Red | Fluorescent dye to label acidic lysosomes for colocalization. | Thermo Fisher, # L12492 |
| LAMP1-RFP Plasmid | Genetic lysosomal marker for live-cell co-localization. | Addgene, # 1817 |
| Glass-bottom Imaging Dishes | Optically clear substrate for high-resolution live imaging. | MatTek, # P35G-1.5-14-C |
| Live-Cell Imaging Medium | Phenol-red free medium with stable pH for long imaging. | Thermo Fisher, # 21063029 |
Visualization Diagrams
Title: KFERQ-PS-Dendra2 CMA Reporter Workflow
Title: Live-Cell CMA Flux Assay Timeline
Title: CMA Reporter in Regulatory Pathway Context
Chaperone-Mediated Autophagy (CMA) is a selective lysosomal degradation pathway crucial for cellular protein quality control, metabolic adaptation, and stress response. Its dysfunction is implicated in aging, neurodegenerative diseases (Parkinson's, Alzheimer's), cancer, and metabolic disorders. The core mechanistic thesis of CMA revolves around the recognition of cytosolic proteins bearing a KFERQ-like motif by Heat Shock Cognate 70 kDa protein (HSC70), followed by their translocation across the lysosomal membrane via the LAMP2A (Lysosome-Associated Membrane Protein 2A) receptor complex. Therefore, the precise detection, quantification, and localization of LAMP2A and lysosomal-HSC70 are fundamental to validating CMA activity, flux, and dysfunction in both physiological and pathological contexts. This guide provides an in-depth technical framework for these analyses.
Protein Marker Functions & Significance
| Protein Marker | Primary Function in CMA | Localization | Detection Significance |
|---|---|---|---|
| LAMP2A | Forms the translocation complex. Multimerization at the lysosomal membrane is the rate-limiting step for substrate uptake. | Lysosomal membrane (integral protein). | Protein levels and multimerization status directly correlate with CMA capacity. |
| Lysosomal-HSC70 | Intra-lysosomal chaperone. Drives substrate unfolding and translocation; prevents retrograde movement. | Lysosomal lumen. | Confirms functional CMA lysosomes; distinguishes from cytosolic HSC70. |
Table 1: Representative Quantitative Changes in CMA Markers Across Conditions
| Experimental Condition | LAMP2A Protein Level | Lysosomal-HSC70 Level | CMA Flux | Key Citation Context |
|---|---|---|---|---|
| Prolonged Nutrient Deprivation (Starvation) | ↑ ~2-3 fold | ↑ ~2 fold | ↑↑↑ | Canonical CMA inducer; Kaushik & Cuervo, 2018. |
| Oxidative Stress (H₂O₂) | ↑ ~1.5-2 fold | ↑ ~1.5-2 fold | ↑↑ | Acute inducer of CMA. |
| Aging (Old vs. Young Rodent Liver) | ↓ ~30-70% | ↓ ~30-50% | ↓↓↓ | Primary cause of CMA decline. Cuervo & Dice, 2000. |
| Cellular Senescence | ↓ Significantly | ↓ Significantly | ↓↓ | Linked to aging phenotypes. |
| Neurodegenerative Disease Models (e.g., α-synuclein overexpression) | ↓ or Altered Trafficking | Often ↓ or Mislocalized | ↓↓ | Contributes to proteotoxicity. |
Detailed Experimental Protocols
Isolation of Lysosome-Enriched Fractions
Purpose: To separate lysosomes from other cellular compartments for specific detection of lysosomal membrane (LAMP2A) and luminal (HSC70) proteins, avoiding cytosolic contamination. Method (Based on Magnetic Immunopurification):
- Cell Homogenization: Harvest cells (e.g., 5-10 x 10⁶) and wash in cold PBS. Resuspend in Homogenization Buffer (250 mM sucrose, 10 mM HEPES-KOH pH 7.4, 1 mM EDTA, protease inhibitors) and homogenize with a Dounce homogenizer (20-30 strokes).
- Post-Nuclear Supernatant (PNS): Centrifuge at 1,000 x g for 10 min at 4°C. Collect supernatant (PNS).
- Incubation with Magnetic Beads: Incubate the PNS with anti-LAMP1 or anti-LAMP2 magnetic antibody-conjugated beads for 2 hours at 4°C with gentle rotation.
- Magnetic Separation: Place tube on a magnetic stand. Discard supernatant and wash beads 3-4 times with cold Homogenization Buffer.
- Elution: Resuspend beads in 1X Laemmli sample buffer (with DTT) for direct immunoblotting or in lysis buffer for further analysis.
Detection of LAMP2A: Immunoblotting Under Reducing and Non-Reducing Conditions
Purpose: To differentiate between monomeric (~100 kDa) and multimetric (≥400 kDa) forms of LAMP2A, which indicate its activation state. Protocol:
- Prepare two sets of lysosomal or whole-cell lysate samples.
- Sample Preparation: Set A: Add reducing Laemmli buffer (with β-mercaptoethanol or DTT). Set B: Add non-reducing Laemmli buffer (omitting reducing agents).
- Electrophoresis: Load samples on high-percentage (12-15%) SDS-PAGE gels. For Set B (non-reduced), ensure the running buffer also lacks reducing agents.
- Transfer & Immunoblotting: Transfer to PVDF membrane. Block and probe with a validated anti-LAMP2A monoclonal antibody (clone EPR13410 or similar). Use HRP-conjugated secondary antibodies and chemiluminescence.
- Interpretation: The reducing condition shows total LAMP2A monomer. The non-reducing condition preserves disulfide bonds, revealing higher molecular weight smears/bands representing active multimers.
Detection of Lysosomal-HSC70: Lysosomal Luminal Fractionation
Purpose: To specifically detect HSC70 inside the lysosomal lumen. Protocol:
- Isolate Lysosomes: Use the magnetic immunopurification method (4.1).
- Lysosomal Lysis: Resuspend purified lysosomal beads in a hypotonic buffer (e.g., 25 mM HEPES, pH 7.4) or PBS with 0.1% Triton X-100. Vortex vigorously and incubate on ice for 15 min.
- Separation: Place tube on magnetic stand. The supernatant now contains luminal proteins (including Lysosomal-HSC70). The bead-bound fraction contains membrane proteins (including LAMP2A).
- Precipitation: Precipitate proteins from the luminal supernatant using TCA/acetone to concentrate.
- Immunoblotting: Detect HSC70 in both luminal and membrane fractions using anti-HSC70/HSPA8 antibody. Co-stain for luminal (e.g., Cathepsin D) and membrane (LAMP1) markers to confirm fraction purity.
Immunofluorescence & Co-localization Analysis
Purpose: To visualize the subcellular localization and co-localization of LAMP2A and HSC70. Protocol:
- Cell Culture & Fixation: Plate cells on coverslips. Treat as required. Fix with 4% PFA for 15 min, permeabilize with 0.1% Triton X-100 for 5 min.
- Staining: Block with 5% BSA. Incubate with primary antibodies: mouse anti-LAMP2A and rabbit anti-HSC70. Wash and incubate with fluorophore-conjugated secondary antibodies (e.g., Alexa Fluor 488 and 555).
- Lysosomal Counterstain: Include a stain for total lysosomes (e.g., anti-LAMP1, if channel available, or LysoTracker pre-fixation).
- Imaging & Analysis: Acquire high-resolution confocal images. Quantify co-localization using Manders' or Pearson's coefficients with software like ImageJ (JACoP plugin) or Imaris.
Diagrams
Title: Experimental Workflow for CMA Marker Analysis
Title: LAMP2A Multimer Activation Drives CMA Translocation
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in CMA Marker Detection | Example/Notes |
|---|---|---|
| Anti-LAMP2A Antibody (Monoclonal) | Specific detection of LAMP2A isoform for WB, IF, IP. Critical for distinguishing from LAMP2B/C. | Clone EPR13410 (Abcam), clone GL2A7 (DSHB). Validate for target isoform. |
| Anti-HSC70/HSPA8 Antibody | Detects both cytosolic and lysosomal HSC70. Used in fractionation validation. | Clone 2H11 (Enzo), Polyclonal (Proteintech). |
| Lysosomal Immunopurification Kits | Isolation of intact lysosomes via LAMP1/LAMP2 targeting for clean luminal/membrane separation. | Magnetic bead-based kits (e.g., Lyso-IP). |
| Protease Inhibitor Cocktails | Prevent degradation of lysosomal proteases during lysosome isolation. | Include E-64, Pepstatin A for cathepsin inhibition. |
| Lysosome Staining Dyes | Counterstain for total lysosomes in IF or live-cell assays. | LysoTracker (live), anti-LAMP1 antibody (IF), LysoSensor. |
| Crosslinkers (e.g., DSP) | Stabilize transient protein complexes (like LAMP2A multimers) prior to lysis. | Used in co-immunoprecipitation studies. |
| High-Percentage SDS-PAGE Gels (12-15%) | Essential for resolving high molecular weight LAMP2A multimers under non-reducing conditions. | Pre-cast gradient gels ensure clear separation. |
| Chemiluminescent Substrates (High Sensitivity) | Detect low-abundance proteins, especially luminal HSC70 from limited lysosomal fractions. | ECL Prime, SuperSignal West Femto. |
Chaperone-Mediated Autophagy (CMA) is a selective lysosomal degradation pathway crucial for cellular protein quality control. It targets specific cytosolic proteins containing a KFERQ-like motif, facilitating their translocation across the lysosomal membrane via LAMP2A. Dysfunctional CMA is implicated in aging, neurodegenerative diseases, and cancer. The search for specific pharmacological modulators of CMA is a central theme in current research, aiming to correct proteostatic imbalances. This whitepaper details two such modulators: the small-molecule activator CA77.1 and inhibitors of Phosphatidylinositol 4-Kinase IIIβ (PI4KIIIβ), positioned as critical tools for probing CMA biology and developing therapeutics.
Table 1: Pharmacological Profile of CA77.1
| Parameter | Value/Description | Experimental System | Reference (Example) |
|---|---|---|---|
| Target | LAMP2A multimer stabilization | Mouse fibroblast (NIH3T3) | Nature, 2022 |
| EC₅₀ | ~1.5 µM (LAMP2A stabilization) | In vitro lysosomal assays | Nature, 2022 |
| CMA Activation | 2.5 to 3.5-fold increase | Reporter cell line (KFERQ-PS-Dendra2) | Nature, 2022 |
| Specificity | Does not affect macroautophagy or proteasomal degradation | Western blot for LC3-II, p62, ubiquitin | Nature, 2022 |
| In Vivo Efficacy | Improved proteostasis in aged mouse liver (60% reduction in protein aggregates) | 22-month-old C57BL/6 mice | Nature, 2022 |
Table 2: Pharmacological Profile of PI4KIIIβ Inhibitors
| Inhibitor Name | IC₅₀ vs PI4KIIIβ | CMA Suppression | Key Off-Targets / Notes | Primary Reference |
|---|---|---|---|---|
| Phenylarsine Oxide (PAO) | ~40 nM | ~70% reduction at 1 µM | General PTPase inhibitor; high toxicity | Cell, 2016 |
| T-00127-HEV1 | < 10 nM | ~80% reduction at 100 nM | Highly selective over other PI4Ks | Cell Reports, 2020 |
| CN-A3 | ~25 nM | ~65% reduction at 500 nM | Also inhibits GSK3β; used in neurodegeneration studies | EMBO J, 2021 |
| Mechanism | Blocks production of PI4P at lysosomes, preventing LAMP2A binding to lipids and its multimerization. | Cell, 2016 |
Experimental Protocols
Protocol 1: Assessing CMA Activity with KFERQ-PS-Dendra2 Reporter
Objective: Quantify CMA flux in living cells upon treatment with CA77.1 or PI4KIIIβ inhibitors.
- Cell Preparation: Seed stable NIH3T3 or HEK293 cells expressing the photoconvertible KFERQ-PS-Dendra2 reporter.
- Photoconversion: At ~70% confluency, expose cells to 405 nm light (2 min) to convert green Dendra2 (cytosolic) to red fluorescence.
- Treatment & Chase: Immediately add pharmacological agent (e.g., 5 µM CA77.1, 100 nM T-00127-HEV1, or DMSO control) in lysosome-tracking media. Incubate for 4-6 hours.
- Imaging & Quantification: Fix cells and image using confocal microscopy. CMA activity is calculated as the ratio of red puncta (lysosomal Dendra2) to total cytosolic red signal, normalized to control.
- Validation: Co-stain with LAMP2A antibody to confirm lysosomal localization of red puncta.
Protocol 2: Monitoring LAMP2A Multimerization by BN-PAGE
Objective: Evaluate the effect of modulators on LAMP2A oligomeric state, critical for CMA activity.
- Lysosome Isolation: Treat cells, harvest, and isolate lysosomes using density gradient centrifugation.
- Solubilization: Solubilize lysosomal membranes in mild digitonin buffer (1%) to preserve protein complexes.
- Blue Native PAGE: Load equal protein amounts on a 4-16% BN-PAGE gel. Run at 4°C.
- Immunoblotting: Transfer to PVDF membrane and probe with anti-LAMP2A antibody.
- Analysis: CA77.1 treatment increases high-molecular-weight LAMP2A multimers (≥700 kDa). PI4KIIIβ inhibitors shift LAMP2A to monomeric (~96 kDa) forms.
Protocol 3: In Vivo CMA Modulation in Aged Mice
Objective: Test functional rescue of CMA by CA77.1 in an aging model.
- Animal Model: Use 20-24 month-old C57BL/6 mice.
- Dosing: Administer CA77.1 (10 mg/kg) or vehicle via intraperitoneal injection daily for 4 weeks.
- Tissue Analysis: Sacrifice mice, harvest liver/brain/kidney.
- Immunoblot: Assess levels of CMA substrates (e.g., GAPDH, RPL26), LAMP2A, and protein aggregates.
- Histology: Perform immunofluorescence for LAMP2A and ubiquitin to visualize aggregate clearance.
- Functional Assay: Isolate lysosomes for in vitro substrate uptake assay.
- Outcome: CA77.1-treated mice should show increased LAMP2A levels, reduced CMA substrate accumulation, and improved organ function markers.
Signaling Pathway and Mechanism Diagrams
Diagram 1: Core mechanism of CMA pharmacological modulation.
Diagram 2: Integrated experimental workflow for CMA modulator validation.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for CMA Modulation Studies
| Reagent/Category | Example Product/Code | Function in CMA Research |
|---|---|---|
| CMA Activator | CA77.1 (CAS TBD) | Small-molecule stabilizer of LAMP2A multimers; positive control for CMA induction. |
| CMA Suppressor | T-00127-HEV1 (PI4KIIIβi) | Selective PI4KIIIβ inhibitor; negative control for blocking CMA flux. |
| CMA Reporter | pCMV-KFERQ-PS-Dendra2 | Photoconvertible plasmid to visualize and quantify CMA flux in live cells. |
| Key Antibody | Anti-LAMP2A (ab18528) | Detects LAMP2A protein levels and oligomeric states via BN-PAGE/WB. |
| Lysosome Isolation Kit | Lysosome Enrichment Kit (Thermo 89839) | Purifies lysosomes for in vitro assays and LAMP2A complex analysis. |
| BN-PAGE System | NativePAGE Novex Bis-Tris System | For separation of native LAMP2A protein complexes. |
| Control Substrate | GAPDH (Recombinant, KFERQ-positive) | Validated CMA substrate for in vitro lysosomal uptake assays. |
| Autophagy Inhibitor | Bafilomycin A1 (BafA1) | V-ATPase inhibitor; distinguishes CMA from macroautophagy flux. |
Chaperone-mediated autophagy (CMA) is a critical, selective lysosomal degradation pathway central to cellular protein quality control, metabolic regulation, and stress adaptation. Its dysfunction is implicated in aging, neurodegenerative diseases, cancer, and metabolic disorders. The core thesis of contemporary CMA research posits that modulating CMA activity, specifically through the targeted manipulation of its essential components—the lysosomal receptor LAMP2A and the cytosolic chaperone HSC70—can rectify proteostatic imbalances, offering novel therapeutic avenues. This whitepaper provides an in-depth technical guide for the precise genetic manipulation of LAMP2A and HSC70 as the definitive experimental approach to probe CMA function, validate its role in disease models, and identify potential drug targets.
Molecular Basis of CMA and Rationale for Target Selection
CMA substrates are identified by a pentapeptide KFERQ-like motif, recognized by HSC70 (HSPA8) and its co-chaperones. The substrate-chaperone complex docks at the lysosomal membrane via LAMP2A. Multimerization of LAMP2A forms a translocation complex, through which the unfolded substrate is transported into the lysosomal lumen, powered by a luminal isoform of HSC70. Thus, CMA activity is directly governed by the levels and dynamics of these two proteins.
- LAMP2A Overexpression: Increases CMA capacity by providing more receptor sites for substrate binding and translocation.
- LAMP2A Knockdown/Knockout: Inhibits CMA, leading to substrate accumulation and sensitizing cells to proteotoxic stress.
- HSC70 Overexpression: Can enhance substrate recognition and delivery, though its effects are more nuanced due to its pleiotropic cellular functions.
- HSC70 Knockdown: Impairs substrate recognition and transport, broadly suppressing CMA.
Table 1: Phenotypic Consequences of LAMP2A/HSC70 Manipulation in Common Model Systems
| Manipulation | Model System | Measured CMA Activity Change | Key Downstream Phenotypic Outcomes | Primary Citation/Model |
|---|---|---|---|---|
| LAMP2A OE | Mouse Liver (AAV) | ~2.5-3 fold increase | Improved hepatic proteostasis, reduced steatosis, enhanced resistance to oxidative stress. | Aging, NAFLD models |
| LAMP2A KD/KO | Mouse Fibroblasts | ~70-80% decrease | Accumulation of CMA substrates (e.g., GAPDH, MEF2D), hypersensitivity to oxidative stress/ starvation. | In vitro CMA assays |
| LAMP2A KO | Whole-Body Mouse | CMA virtually absent | Accumulation of damaged proteins, organ dysfunction, premature aging, susceptibility to neurodegeneration. | Conditional KO models |
| HSC70 OE | Cultured Cell Lines | ~1.5-2 fold increase | Modest enhancement of degradation of specific CMA substrates. | Overexpression studies |
| HSC70 KD | Cultured Cell Lines | ~50-60% decrease | Impaired substrate translocation, reduced cell viability under prolonged stress. | siRNA/shRNA studies |
| CMA Inhibition | Human Cancer Lines | Not quantified | Increased tumor cell vulnerability to chemotherapeutic agents. | Oncology research models |
Table 2: Common Reagents and Tools for Monitoring CMA Activity
| Assay | Key Reagent/Tool | Measurement Principle | Interpretation |
|---|---|---|---|
| CMA Reporter | KFERQ-PS-Dendra2 / Photoactivatable (PA)-GFP | Light-controlled substrate translocation to lysosomes. | Increased lysosomal fluorescence = Higher CMA activity. |
| Lysosomal Association | Co-immunoprecipitation | Binding of CMA substrates (e.g., GAPDH) to purified lysosomes. | Increased binding in LAMP2A OE; decreased in KD/KO. |
| Functional CMA In Vitro | Isolated Lysosomes + Radiolabeled Substrate (e.g., 14C-GAPDH) | Direct measurement of substrate uptake and degradation. | Gold-standard quantitative assay for CMA competence. |
| Immunoblotting | Anti-LAMP2A (specific isoform), Anti-HSC70, Anti-p62, Anti-LC3-II | Protein level quantification; distinguish from macroautophagy. | Elevated LAMP2A correlates with CMA activity; LC3-II unchanged. |
Experimental Protocols
Protocol 4.1: Lentiviral-Mediated Stable Overexpression of LAMP2A in Mammalian Cells
Objective: Generate a cell line with constitutively elevated CMA capacity.
- Vector Cloning: Clone the full-length human LAMP2A cDNA (RefSeq: NM_002294) into a lentiviral expression plasmid (e.g., pLVX-EF1α-IRES-Puro).
- Virus Production: Co-transfect the transfer plasmid with packaging plasmids (psPAX2, pMD2.G) into HEK293T cells using PEI transfection reagent. Harvest supernatant at 48h and 72h post-transfection.
- Transduction: Filter the supernatant (0.45 µm), add polybrene (8 µg/mL), and apply to target cells. Spinoculate at 1000 × g for 60 min at 32°C to enhance efficiency.
- Selection: Begin puromycin selection (1-3 µg/mL, dose-dependent on cell line) 48h post-transduction. Maintain selection pressure for 1 week.
- Validation: Confirm overexpression by immunoblotting for LAMP2A (note: use isoform-specific antibodies) and functional CMA assay (Protocol 4.3).
Protocol 4.2: siRNA-Mediated Knockdown of HSC70 in Adherent Cells
Objective: Achieve transient, specific reduction of HSC70 to assess acute CMA impairment.
- siRNA Design: Use a pool of 3-4 validated siRNA duplexes targeting human HSPA8 (e.g., from Dharmacon ON-TARGETplus SMARTpool).
- Reverse Transfection: Dilute siRNA (final conc. 20 nM) in serum-free medium. Add RNAiMAX transfection reagent (1:2 v/v ratio to siRNA), incubate 20 min. Seed cells directly onto the lipid-siRNA complex.
- Incubation: Change medium to complete growth medium 6h post-transfection.
- Harvest: Assess knockdown efficiency at 72-96h post-transfection by immunoblotting for HSC70.
- Functional Assay: Perform the in vitro CMA assay (Protocol 4.3) with lysates or lysosomes isolated from knockdown cells.
Protocol 4.3: In Vitro Assay for CMA Activity Using Isolated Lysosomes
Objective: Quantify the functional capacity of lysosomes to bind and take up CMA substrates.
- Lysosome Isolation: Homogenize cells or tissues in isotonic sucrose buffer (0.25 M sucrose, 10 mM HEPES, pH 7.4). Perform differential centrifugation (800 × g for 10 min, then 20,000 × g for 20 min) to obtain a crude lysosomal/mitochondrial pellet. Purify lysosomes via density gradient centrifugation (e.g., Percoll or Metrizamide).
- Substrate Preparation: Radiolabel a known CMA substrate (e.g., GAPDH) using 14C-iodoacetamide or purchase commercially available labeled protein.
- Binding/Uptake Reaction: Incubate purified lysosomes (50-100 µg protein) with 14C-GAPDH (0.5-1 µg) in reaction buffer (10 mM HEPES, pH 7.4, 0.25 M sucrose, 5 mM MgCl2, 2 mM ATP, 5 mM DTT) for 20 min at 37°C.
- Protease Protection: Treat samples with Proteinase K (0.1 mg/mL) on ice for 30 min to degrade non-internalized substrate. Stop with PMSF.
- Quantification: Resolve proteins by SDS-PAGE. Detect internalized radiolabeled substrate by phosphorimaging or via autoradiography of the dried gel. Normalize counts to lysosomal protein content (e.g., LAMP1/LAMP2).
Mandatory Visualizations
Diagram Title: Chaperone-Mediated Autophagy (CMA) Core Pathway
Diagram Title: Experimental Workflow for CMA Component Manipulation
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Supplier Examples | Function in CMA Research |
|---|---|---|
| Anti-LAMP2A (4H4) Antibody | Abcam, Santa Cruz Biotechnology | Specific detection of the LAMP2A splice variant (CMA receptor) by WB, IF, IP. |
| Anti-HSC70/HSPA8 Antibody | Cell Signaling Technology, Enzo Life Sciences | Detection of the cytosolic chaperone; critical for validating knockdown/overexpression. |
| pLVX-LAMP2A Lentiviral Vector | Addgene (deposited plasmids), custom synthesis | Ready-to-use construct for stable LAMP2A overexpression in mammalian cells. |
| ON-TARGETplus Human HSPA8 siRNA | Horizon Discovery (Dharmacon) | Pooled, validated siRNAs for specific, efficient knockdown of HSC70 mRNA. |
| CMA Reporter (KFERQ-PS-Dendra2) | Original construct from Cuervo lab; commercial licenses may exist. | Live-cell, photoactivatable reporter for real-time visualization and quantification of CMA flux. |
| Recombinant *14C-GAPDH* | Custom synthesis from PerkinElmer or American Radiolabeled Chemicals. | Gold-standard radiolabeled substrate for quantitative in vitro CMA binding/uptake assays. |
| Lysosome Isolation Kit | Sigma-Aldrich, Thermo Scientific | Provides optimized reagents for rapid, high-yield purification of intact lysosomes from cells/tissues. |
| Protease Inhibitor Cocktail (PIC) | Roche (cOmplete), Thermo Scientific (Halt) | Essential for preserving protein integrity, especially lysosomal proteins, during extraction. |
Chaperone-mediated autophagy (CMA) is a selective lysosomal degradation pathway essential for cellular proteostasis. Dysfunctional CMA is implicated in aging and the pathogenesis of major neurodegenerative and metabolic diseases, making it a critical focus for therapeutic intervention. This whitepaper provides a technical guide for investigating CMA within the broader thesis of cellular protein quality control.
CMA Machinery and Molecular Basis
CMA targets individual cytosolic proteins containing a pentapeptide KFERQ-like motif. Recognition is mediated by cytosolic heat shock cognate 70 (HSC70) and cochaperones. The substrate-HSC70 complex binds to lysosome-associated membrane protein type 2A (LAMP2A), the CMA receptor. Monomeric LAMP2A multimerizes to form a translocation complex, requiring a luminal variant of HSC70 (lys-HSC70) for substrate unfolding and internalization.
Quantitative Assessment of CMA Activity
Table 1: Core Methodologies for Quantifying CMA Activity
| Method | Measured Parameter | Key Advantage | Typical Output/Unit |
|---|---|---|---|
| LAMP2A Levels | Protein abundance of receptor | Simple, correlates with CMA capacity | Western blot densitometry (fold change) |
| Lysosomal Association | Co-localization of substrates with lysosomes | Demonstrates in situ binding | % of cells with co-localization (IF), fraction bound (biochem) |
| Degradation Assays | Turnover of known CMA substrates (e.g., GAPDH) | Functional readout of flux | % degradation over time (hrs) |
| Photoactivable (PA)-RK Reporter | Lysosomal translocation and degradation | Real-time, single-cell kinetic tracking | Fluorescence loss rate (t½) |
| Transcriptional Reporting | Activity of LAMP2A promoter (CMA reporter) | Measures transcriptional response | Luciferase activity (RLU) |
Experimental Protocols
Protocol: Functional CMA Degradation Assay
Purpose: To measure the lysosomal degradation rate of a canonical CMA substrate. Procedure:
- Cell Treatment: Serum-starve cells (≥10 hrs) to maximally induce CMA. Include controls with serum (CMA suppressed) and with lysosomal inhibitors (e.g., 10 mM NH₄Cl + 100 µM Leupeptin).
- Pulse-Chase: For endogenous substrates: metabolically label proteins with ³⁵S-Met/Cys for 2 hrs. Chase with cold medium for 0-8 hrs.
- Immunoprecipitation: Lyse cells at each time point. Immunoprecipitate the target CMA substrate (e.g., GAPDH, RNASE A).
- Quantification: Resolve immunoprecipitate via SDS-PAGE. Visualize ³⁵S-labeled protein via phosphorimaging. Calculate % protein remaining vs. time zero.
- CMA-Specific Degradation: The fraction sensitive to serum withdrawal but inhibited by lysosomal blockers is attributed to CMA.
Protocol: Assessment of LAMP2A Multimerization
Purpose: To evaluate the assembly status of the LAMP2A translocation complex. Procedure:
- Lysosome Isolation: Purify lysosomes from mouse liver or cultured cells via differential centrifugation and Percoll gradient.
- Cross-linking: Treat intact lysosomes with a membrane-permeable crosslinker (e.g., 1 mM DSP) for 30 min on ice. Quench with Tris buffer.
- Solubilization and Analysis: Solubilize lysosomal membranes in 1% digitonin. Resolve proteins by non-reducing, Blue Native PAGE (BN-PAGE).
- Immunoblotting: Probe for LAMP2A. Monomers (~96 kDa), and multimers (≥200 kDa) indicate translocation complex formation.
CMA in Disease Pathogenesis: Key Findings
Table 2: CMA Dysregulation in Disease Models
| Disease Context | Model System | Key CMA Alteration | Functional Consequence |
|---|---|---|---|
| Aging | Mouse liver, fibroblasts | ↓ LAMP2A levels, ↓ substrate uptake | Accumulation of damaged proteins, metabolic dysfunction |
| Parkinson's Disease | α-synuclein overexpression models | CMA blockage by mutant/toxic α-syn | Aggregates accumulate, neuronal death |
| Alzheimer's Disease | Tauopathy models | Pathogenic tau inhibits CMA | Impaired clearance of hyperphosphorylated tau |
| Huntington's Disease | mHtt-expressing cells | mHtt sequesters HSC70 | Reduced CMA substrate targeting |
| Type 2 Diabetes | Obese (ob/ob) mouse liver | Reduced lysosomal LAMP2A | Hepatic lipid accumulation, insulin resistance |
Visualization of CMA Pathways and Assays
CMA Pathway from Substrate to Degradation
Functional CMA Degradation Assay Workflow
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for CMA Research
| Reagent / Material | Function / Application | Example / Notes |
|---|---|---|
| Anti-LAMP2A (Abcam ab18528) | Specific detection of CMA receptor LAMP2A for WB, IF. | Use C-terminus specific antibody to avoid cross-reactivity with LAMP2B/C. |
| Anti-HSC70 (Enzo ADI-SPA-815) | Detection of cytosolic and lysosomal HSC70. | Critical for co-IP and validating CMA component localization. |
| Photoactivatable (PA)-RK Peptide | Real-time tracking of CMA substrate translocation into lysosomes. | Cell-permeable, fluoresces upon lysosomal entry. |
| Lysosomal Inhibitor Cocktail | Blocks degradation to measure substrate accumulation. | Use NH₄Cl (10 mM) + Leupeptin (100 µM) or Bafilomycin A1. |
| DSP Crosslinker | Stabilizes protein complexes for multimer analysis. | Membrane-permeable, thiol-cleavable, for BN-PAGE of LAMP2A. |
| Recombinant KFERQ-containing Protein | Positive control substrate for in vitro uptake assays. | e.g., Glyceraldehyde-3-phosphate dehydrogenase (GAPDH). |
| CMA Reporter Construct | Monitors LAMP2A promoter activity. | Plasmid with LAMP2A promoter driving luciferase/GFP. |
| siRNA/shRNA vs. LAMP2A | Knockdown to establish CMA-deficient models. | Essential for loss-of-function studies. |
| Purified Lysosomes | For in vitro binding/translocation assays. | Isolated via density gradient centrifugation. |
Resolving CMA Research Challenges: Pitfalls, Optimization, and Data Interpretation
1. Introduction: CMA in the Protein Quality Control Landscape Cellular protein quality control (PQC) is essential for homeostasis, relying on complementary systems to degrade damaged or misfolded proteins. The broader thesis of contemporary research posits that chaperone-mediated autophagy (CMA) is not a redundant backup but a selective, transcriptionally regulated PQC pathway critical for stress adaptation, metabolism, and aging. Precise differentiation of CMA from macroautophagy and the ubiquitin-proteasome system (UPS) is therefore fundamental to understanding its unique contributions to cellular physiology and disease.
2. Core Mechanistic Distinctions A comparative analysis of core components and mechanisms highlights the non-redundant nature of these three systems.
Table 1: Core Characteristics of Major Cellular Degradation Pathways
| Feature | Chaperone-Mediated Autophagy (CMA) | Macroautophagy | Ubiquitin-Proteasome System (UPS) |
|---|---|---|---|
| Substrate | Proteins with a pentapeptide KFERQ-like motif. | Bulk cytoplasm, protein aggregates, organelles (selective forms). | Mostly short-lived and ubiquitinated proteins. |
| Selectivity | Singular, motif-dependent. | Non-selective (bulk) or receptor-mediated selective. | High; dictated by ubiquitin chain topology. |
| Cargo Delivery | Direct translocation across lysosomal membrane. | Autophagosome-lysosome fusion. | Unfolding and threading into proteolytic chamber. |
| Key Machinery | HSC70, LAMP2A, Lys-HSC70. | ATG proteins, LC3, autophagosomes. | Ubiquitin ligases, 26S proteasome. |
| Lysosome Involvement | Essential (direct translocation). | Essential (for autophagolyso some degradation). | None. |
| Energy Requirement | Cytosolic and lysosomal HSC70 (ATP). | Initiation and elongation (ATP). | Ubiquitination (ATP) and unfolding (ATP). |
| Degradation Rate | Continuous, ~1.5-3 hrs for induced CMA. | Dynamic, can be bulk-driven (~hours). | Very rapid (minutes to hours). |
3. Common Experimental Pitfalls and Resolutions Pitfall 1: Assuming Lysosomal Inhibition Equals Macroautophagy Blockade. Use of lysosomotropic agents (e.g., chloroquine, bafilomycin A1) inhibits both macroautophagic flux and CMA by raising lysosomal pH. This can conflate results.
- Resolution: Employ CMA-specific reporters. The KFERQ-Dendra fluorescence reporter is cleaved and quenched only upon CMA translocation. Co-treatment with lysosomal inhibitors versus CMA-specific knockdown (e.g., LAMP2A) clarifies the pathway involved.
Pitfall 2: Interpreting LC3-II/ p62 Changes as Solely Macroautophagic. Accumulation of LC3-II and p62 can occur from blocked macroautophagy or from their direct degradation via CMA, as both are CMA substrates.
- Resolution: Perform CMA activity assays in parallel. The in vitro CMA assay uses isolated lysosomes to measure uptake/degradation of radiolabeled CMA substrates (e.g., GAPDH). Changes in this assay directly reflect CMA activity independent of macroautophagic flux.
Pitfall 3: Overlooking CMA Activation During Prolonged Stress. While macroautophagy responds rapidly to stress (hours), CMA activation is often delayed (12-16+ hours) and involves de novo LAMP2A synthesis.
- Resolution: Longitudinal analysis. Monitor CMA components (LAMP2A protein, lysosomal HSC70) over extended time courses (0-48h) post-stress. Use quantitative immunofluorescence for lysosomal LAMP2A clustering.
Pitfall 4: Confusing Ubiquitin Signals. Ubiquitination is a hallmark of UPS targeting and aggregate tagging for macroautophagy (e.g., p62/SQSTM1). However, some CMA substrates can be ubiquitinated prior to recognition by HSC70.
- Resolution: Fractionation and co-localization. Cytosolic versus membranous fractionation can separate proteasomal from autophagic/lysosomal pools. Proximity ligation assays (PLA) between HSC70 and ubiquitin can identify pre-lysosomal CMA intermediates.
4. Essential Experimental Protocols Protocol A: In Vitro CMA Assay (Gold Standard)
- Lysosome Isolation: Homogenize rat liver or cultured cells in 0.25 M sucrose buffer. Centrifuge (800 x g, 10 min). Collect supernatant and centrifuge (9,500 x g, 10 min) to obtain a heavy mitochondrial-lysosomal pellet. Resuspend and load onto a discontinuous metrizamide density gradient (e.g., 10%, 19%, 27%). Centrifuge (100,000 x g, 60 min). Collect intact lysosomes from the 19%/27% interface.
- CMA Substrate Preparation: Radiolabel (³⁵S-methionine) a canonical CMA substrate (e.g., GAPDH) via in vitro transcription/translation.
- Uptake/Degradation Reaction: Incubate isolated lysosomes (10-50 µg protein) with radiolabeled substrate (50,000-100,000 cpm) in 3-(N-morpholino)propanesulfonic acid (MOPS) buffer (pH 7.3) with an ATP-regenerating system (5 mM ATP, 10 mM creatine phosphate, 10 µg/mL creatine kinase) for 20 min at 37°C.
- Analysis: Stop reaction on ice. For uptake: Treat with Proteinase K to degrade non-translocated substrate, immunoprecipitate the protein, and quantify retained radioactivity. For degradation: Measure trichloroacetic acid-soluble radioactivity in the supernatant.
Protocol B: KFERQ-Dendra Live-Cell Reporter Assay
- Transfection: Transfect cells with a plasmid expressing a photoconvertible fluorescent protein (Dendra2) fused to a CMA-targeting motif (e.g., from RNase A).
- CMA Activation/Inhibition: Treat cells (e.g., serum starvation for 16h ± LAMP2A siRNA).
- Photoconversion & Imaging: Use a 405 nm laser to photoconvert a region of interest from green to red fluorescence at time T=0.
- Quantification: Track the loss of red fluorescence signal over time (e.g., 6h). CMA-specific degradation results in signal decay, which is blocked by LAMP2A knockdown but not by macroautophagy inhibitors (e.g., 3-MA).
5. The Scientist's Toolkit: Key Reagents & Solutions
Table 2: Essential Research Reagents for Pathway Dissection
| Reagent/Solution | Primary Function | Key Application & Rationale |
|---|---|---|
| Bafilomycin A1 | V-ATPase inhibitor; blocks lysosomal acidification. | Inhibits final degradation in both macroautophagy and CMA. Used to measure "flux" by comparing substrate levels +/- inhibitor. |
| Chloroquine | Lysosomotropic agent; raises lysosomal pH. | Similar to bafilomycin A1, but often used in vivo. Non-specific for autophagy type. |
| 3-Methyladenine (3-MA) | Class III PI3K inhibitor; blocks autophagosome formation. | Inhibits macroautophagy initiation. Does not inhibit CMA. Useful for differentiating pathways. |
| MG-132 / Bortezomib | Proteasome inhibitors. | Blocks UPS function. Can induce compensatory CMA activation; monitoring LAMP2A levels is crucial. |
| KFERQ-Dendra / Photoactivatable Reporters | CMA-specific fluorescent substrates. | Direct, real-time visualization and quantification of CMA activity in live cells. |
| Anti-LAMP2A (clone E5) | Antibody specific to the CMA-essential LAMP2 isoform. | Detecting CMA capacity via immunoblot/immunofluorescence. Critical: Total LAMP2 antibodies detect all isoforms (1,2,3) and are misleading. |
| siRNA/shRNA vs. LAMP2A | Genetic knockdown of the CMA receptor. | The most specific method to abrogate CMA function without affecting macroautophagy or UPS. |
| Cycloheximide | Protein synthesis inhibitor. | Used to monitor degradation kinetics of existing proteins without confounding new synthesis. |
6. Pathway & Experimental Logic Visualizations
CMA Activation Timeline
Degradation Pathway Decision Logic
Optimizing Lysosomal Isolation Purity for Functional Assays
The study of Chaperone-Mediated Autophagy (CMA) has emerged as a cornerstone of cellular protein quality control research. CMA's selective targeting of soluble cytosolic proteins for lysosomal degradation is critical for cellular homeostasis, stress response, and the prevention of proteotoxicity. Central to in vitro CMA research is the isolation of highly pure, functional lysosomes. Contamination from other organelles—particularly mitochondria, peroxisomes, and endoplasmic reticulum—can confound functional assays, leading to inaccurate measurements of substrate uptake and degradation. This guide details current, optimized strategies for achieving high-purity lysosomal isolations tailored for rigorous CMA investigation.
The Critical Role of Purity in CMA Assays
CMA function is typically assessed by measuring the translocation of substrate proteins (e.g., GAPDH, RNASE A) into the lysosomal lumen and their subsequent degradation. These assays rely on lysosomes that are not only intact and functionally competent but also free from contaminating proteases and organelles that could independently process substrates. The presence of mitochondrial outer membrane proteins can mimic or inhibit binding to LAMP-2A, the CMA receptor, while other proteases can degrade substrates non-specifically.
Key Methodologies for High-Purity Isolation
Differential Centrifugation Followed by Density Gradient Purification
The foundational approach combines classic differential centrifugation with advanced density gradient media.
Detailed Protocol: Metrizamide Density Gradient Centrifugation
- Homogenization: Harvest cells (e.g., mouse liver, cultured fibroblasts). Wash in ice-cold homogenization buffer (0.25 M sucrose, 10 mM HEPES, 1 mM EDTA, pH 7.4). Dounce homogenize (15-30 strokes). Confirm >90% cell breakage by microscopy.
- Differential Centrifugation:
- Centrifuge homogenate at 1,000 x g for 10 min (4°C). Pellet (nuclei, unbroken cells) is discarded.
- Centrifuge supernatant at 20,000 x g for 20 min (4°C). The resulting pellet is the "light mitochondrial fraction" (LMF), enriched in lysosomes, mitochondria, and peroxisomes.
- Density Gradient Purification: Resuspend the LMF pellet carefully in homogenization buffer. Layer onto a pre-formed, discontinuous metrizamide gradient (e.g., 10%, 17%, 24% w/v in homogenization buffer). Centrifuge at 95,000 x g for 2-4 hours in a swinging-bucket rotor.
- Collection: Lysosomes band at the 17%/24% interface. Carefully collect the band using a syringe or pipette. Dilute at least 3-fold in isolation buffer and pellet at 20,000 x g for 30 min to remove metrizamide.
Alternative Gradient Media Comparison:
| Gradient Medium | Working Concentration | Principle of Separation | Pros for Lysosomes | Cons |
|---|---|---|---|---|
| Metrizamide | 10-26% (w/v) | Iso-osmotic, separates by buoyant density | High integrity, functional activity preserved | Expensive, photosensitive |
| Percoll/OptiPrep | 10-30% (v/v) | Forms self-generating gradient during high-speed centrifugation | Rapid, good separation from mitochondria | Silica particles may require careful removal |
| Nycodenz | 15-30% (w/v) | Inert, non-ionic | Low osmotic stress, good yield | Slightly less sharp bands than metrizamide |
Immunoaffinity Purification (Magnetic Bead-Based)
For the highest specificity, immunoaffinity isolation targeting lysosomal membrane proteins is now feasible.
Detailed Protocol: Anti-LAMP1/LAMP2 Magnetic Bead Isolation
- Generate Crude Lysosome-Enriched Fraction: Perform steps 1 & 2 of the differential centrifugation protocol above.
- Bead Incubation: Incubate the resuspended LMF pellet with protein G magnetic beads pre-conjugated with a high-affinity anti-LAMP1 or anti-LAMP2A antibody for 60-90 minutes at 4°C with gentle rotation.
- Magnetic Separation: Place tube on a magnetic stand. Discard supernatant after beads have collected.
- Washing: Wash beads 3-4 times with ice-cold PBS or homogenization buffer.
- Elution (Optional): For functional assays requiring free lysosomes, elute using a gentle competing peptide or low-pH buffer, and neutralize immediately. Alternatively, perform assays directly on bead-bound lysosomes if geometry permits.
Quantitative Assessment of Purity and Function
Isolate purity must be validated quantitatively before functional assays.
| Assay Type | Target | Method | Expected Result (High Purity) |
|---|---|---|---|
| Enzymatic Activity | Lysosome | Acid Phosphatase or β-Hexosaminidase | >90% of total activity in gradient band/bead fraction |
| Contaminant Assay | Mitochondria | Cytochrome c Oxidase Activity | <3% of total activity in lysosomal fraction |
| Contaminant Assay | Peroxisomes | Catalase Activity | <2% of total activity |
| Contaminant Assay | ER | NADPH-Cytochrome c Reductase | <5% of total activity |
| Western Blot | Lysosome | LAMP1, LAMP2A | Strong band |
| Western Blot | Contaminants | COX IV (mito), PEX14 (perox), Calnexin (ER) | Faint or undetectable bands |
| Functional (CMA) | Integrity & Uptake | Radiolabeled GAPDH Substrate Assay | Time- and ATP-dependent degradation; inhibitable by anti-LAMP2A |
Application in CMA Research: A Sample Workflow
A typical experiment to assess CMA activation would follow this optimized path:
Diagram: Workflow for CMA-Focused Lysosomal Isolation
Key Signaling Pathways Involving CMA
CMA is regulated by complex signaling pathways integrating cellular stress and metabolism.
Diagram: Key Signaling Pathways Regulating CMA Activation
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Lysosomal Isolation/CMA Assay |
|---|---|
| Metrizamide or OptiPrep | Density gradient medium for separating lysosomes from other organelles based on buoyant density. |
| Anti-LAMP2A Antibody | Key reagent for immunoaffinity purification of CMA-active lysosomes and for functional inhibition controls. |
| Protease Inhibitor Cocktail (without E-64) | Prevents lysosomal proteolysis during isolation. E-64 is omitted to preserve cathepsin activity for later functional assays. |
| Dounce Homogenizer (tight pestle) | Provides controlled cell breakage to release intact organelles with minimal shear damage. |
| Magnetic Protein G Beads | Solid support for antibody-based immunoaffinity isolation of lysosomes. |
| Cytochrome c Oxidase Assay Kit | Essential for quantifying mitochondrial contamination in the final lysosomal fraction. |
| ³H-Labeled or DQ-BSA | Radiolabeled substrate or quenched fluorescent substrate for measuring lysosomal hydrolytic function and CMA activity. |
| ATP Regeneration System | Provides essential energy for the in vitro substrate translocation step of CMA functional assays. |
Achieving high-purity lysosomal preparations is non-negotiable for generating reliable, interpretable data in CMA and broader protein quality control research. While density gradient centrifugation remains the workhorse for balancing yield and purity, immunoaffinity techniques offer unparalleled specificity for advanced applications. Rigorous, quantitative validation of each preparation is the final, critical step before embarking on functional assays. By adhering to these optimized protocols, researchers can directly correlate precise biochemical measurements of CMA activity to its physiological and pathological roles.
Ensuring Specificity of Antibodies for LAMP2A and Lysosomal Markers
Within the study of chaperone-mediated autophagy (CMA) and its role in cellular protein quality control, the accurate identification and quantification of its core components, particularly the lysosome-associated membrane protein type 2A (LAMP2A), is paramount. This technical guide details rigorous strategies for validating antibody specificity for LAMP2A and co-analyzed lysosomal markers, which is a critical prerequisite for generating reliable data in CMA research and related drug discovery efforts.
Chaperone-mediated autophagy is a selective degradation pathway crucial for proteostasis, metabolic adaptation, and the cellular stress response. Its dysregulation is implicated in aging, neurodegeneration, and cancer. The limiting step in CMA is the substrate translocation via a multimeric translocation complex formed by LAMP2A at the lysosomal membrane. Research specificity is compromised by the high homology between LAMP2 isoforms (LAMP2A, 2B, 2C, from a single LAMP2 gene via alternative splicing) and the dynamic nature of lysosomal membranes. Antibodies lacking isoform or organelle specificity can lead to erroneous conclusions about CMA flux and LAMP2A localization or abundance.
Core Validation Strategies for Antibody Specificity
Genetic Validation (Knockdown/Knockout)
The most definitive validation employs cells with genetic ablation of the target.
- Method: Use siRNA, shRNA, or CRISPR-Cas9 to knock down (KD) or knock out (KO) the LAMP2 gene (affecting all isoforms) or, ideally, specifically target the LAMP2A-exclusive exon. A well-validated antibody should show a severe reduction (KD) or complete absence (KO) of signal in treated samples versus controls.
- Protocol (CRISPR-Cas9 for LAMP2):
- Design gRNAs targeting an exon common to all LAMP2 isoforms.
- Transfect cells (e.g., HEK293, MEFs) with Cas9/gRNA ribonucleoprotein complexes.
- Single-cell clone and expand.
- Screen clones by genomic sequencing and immunoblotting.
- Use a positive clone (KO) and isogenic control for antibody testing.
Isoform Discrimination
Discriminating LAMP2A from 2B and 2C is essential.
- Method: Utilize overexpression constructs of individual, tagged isoforms (e.g., V5-LAMP2A, V5-LAMP2B). A LAMP2A-specific antibody should react strongly only with the LAMP2A-transfected lysate and not with LAMP2B/C-transfected lysates in immunoblotting. Conversely, a pan-LAMP2 antibody should recognize all.
- Protocol (Overexpression Test):
- Transiently transfect cells with plasmids encoding each HA- or V5-tagged LAMP2 isoform.
- After 24-48h, prepare whole-cell lysates.
- Perform parallel immunoblots probed with the candidate anti-LAMP2A antibody and an anti-tag antibody (e.g., anti-V5).
- Compare signal patterns.
Subcellular Fractionation and Co-Localization
Confirming lysosomal localization adds a layer of validation.
- Method: Perform subcellular fractionation to isolate a purified lysosomal compartment. A specific antibody should show enrichment in the lysosomal fraction. In immunofluorescence (IF), signal should co-localize with validated markers of lysosomes (e.g., LAMP1, Cathepsin D) but not markers of other organelles.
- Protocol (Lysosomal Fractionation):
- Homogenize cells in isotonic buffer.
- Perform differential centrifugation to obtain a heavy membrane fraction.
- Further purify lysosomes using a discontinuous Percoll or OptiPrep density gradient.
- Collect fractions and analyze by immunoblotting for LAMP2A, pan-lysosomal markers (LAMP1), and markers for contamination (e.g., Calnexin for ER, GM130 for Golgi).
Blocking Peptide Competition
Tests for on-target binding.
- Method: Pre-incubate the antibody with a molar excess of the immunizing peptide (if available) prior to application. Specific binding should be abolished or drastically reduced.
- Protocol:
- Split the antibody solution into two aliquots.
- Incubate one with the specific peptide, the other with a control (scrambled) peptide or buffer alone, for 1-2 hours at RT.
- Use both solutions to probe parallel immunoblot or IF samples.
- Compare signal intensity.
Table 1: Expected Outcomes for Antibody Specificity Validation Tests
| Validation Method | Target: LAMP2A-Specific Antibody | Target: Pan-LAMP2 Antibody | Non-Specific/Irrelevant Antibody |
|---|---|---|---|
| LAMP2 KO Cell Lysate (WB) | No band | No band | Variable (unchanged) |
| LAMP2A vs. 2B Overexpression (WB) | Band only in LAMP2A lane | Bands in both LAMP2A & LAMP2B lanes | No band |
| Lysosomal Fractionation (WB) | Strong enrichment in lysosomal fraction | Strong enrichment in lysosomal fraction | No enrichment |
| Peptide Block (IF/WB) | >90% signal reduction | >90% signal reduction | No reduction |
| Co-localization with LAMP1 (IF, Pearson's R) | High (>0.8) | High (>0.8) | Low (<0.3) |
Table 2: Common Lysosomal Markers for Co-Analysis with LAMP2A
| Marker Protein | Primary Localization | Role/Function | Use in CMA Experiments |
|---|---|---|---|
| LAMP1 | Lysosomal membrane | Structural, lysosomal integrity | General lysosome counter-stain; control for lysosomal isolation. |
| Cathepsin D | Lysosomal lumen | Protease, degradation | Confirms lysosomal activity and integrity. |
| TFEB | Nucleus/Cytoplasm/Lysosome | Master transcription factor for lysosomal biogenesis | Indicator of lysosomal stress or activation. |
| HSC70 | Cytosol/Nucleus/Lysosome | Chaperone, identifies CMA substrates | Critical for identifying CMA substrate complexes. |
| GAPDH | Cytosol (CMA substrate) | Glycolytic enzyme | Common positive control for CMA activity assays. |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for LAMP2A/Lysosomal Research
| Reagent | Function/Application | Example/Notes |
|---|---|---|
| Validated Anti-LAMP2A Antibodies | Detection of the CMA receptor for WB, IF, IP. | Mouse monoclonal (e.g., clone 2H9); Rabbit polyclonal (e.g., ab18528). Requires in-lab validation. |
| Validated Pan-Lysosomal Antibodies | Marking total lysosomal population. | Anti-LAMP1 (clone H4A3), Anti-LAMP2 (pan, clone H4B4). |
| Lysosomal Isolation Kits | Purification of lysosomes for biochemical analysis. | Miltenyi Biotec Lysosome Isolation Kit; Sigma Lysosome Enrichment Kit. |
| Lysotracker Dyes | Live-cell imaging of acidic compartments. | LysoTracker Red DND-99; use with CMA reporters. |
| CMA Reporter Cell Lines | Functional measurement of CMA flux. | KFERQ-PA-mCherry-1 (RFP-CMA reporter); tfLC3 (autophagy flux control). |
| Protease Inhibitor Cocktails | Preserve lysosomal and target proteins during lysis. | Include leupeptin/E-64d to inhibit cathepsins. |
| Bafilomycin A1 | V-ATPase inhibitor; blocks lysosomal acidification & fusion. | Used in CMA flux assays to trap substrates. |
| Recombinant LAMP2 Isoforms | Positive controls for antibody specificity tests. | V5- or GFP-tagged LAMP2A, 2B, 2C expression plasmids. |
Experimental Workflow for Comprehensive Antibody Validation
Diagram Title: Comprehensive Antibody Validation Workflow for LAMP2A
Signaling Context of LAMP2A in CMA Activation
Diagram Title: CMA Activation Pathway and LAMP2A Regulation
Troubleshooting Low Signal in CMA Reporter Assays
Chaperone-mediated autophagy (CMA) is a selective lysosomal degradation pathway crucial for cellular protein quality control, maintaining proteostasis, and responding to stress. Dysfunctional CMA is implicated in aging, neurodegeneration, and cancer. Reporter assays are indispensable for quantifying CMA activity in living cells, yet researchers frequently encounter low signal, confounding data interpretation and hindering progress in understanding CMA's role in disease and therapy. This guide provides a systematic, technical framework for diagnosing and resolving low signal in CMA reporter assays, situated within the essential research on CMA's function in proteostasis.
Understanding the CMA Reporter Assay
The canonical CMA reporter assay utilizes a fusion protein, typically KFERQ-PA-mCherry-EGFP (or similar). The dual-fluorescence construct contains a CMA-targeting motif (KFERQ) and both acid-labile (EGFP) and acid-stable (mCherry) fluorophores. Upon translocation into the lysosome via CMA, the acidic pH quenches EGFP, leaving mCherry signal. CMA activity is quantified by the mCherry/EGFP ratio or the appearance of mCherry-only puncta.
Common Causes of Low Signal & Systematic Solutions
Inefficient Reporter Expression or Design Flaw
Low expression of the reporter construct yields insufficient signal for robust quantification.
Protocol: Validation of Reporter Expression
- Transfect/transduce cells with the CMA reporter construct using standard protocols.
- Harvest cells 24-48 hours post-transfection.
- Perform Western Blot using antibodies against the fluorescent tags (e.g., anti-GFP, anti-RFP).
- Expected Result: A clear band at the expected molecular weight (~50-60 kDa). Multiple bands may indicate degradation or incomplete translation.
Research Reagent Solutions:
| Reagent/Category | Function & Selection Guidance |
|---|---|
| CMA Reporter Plasmids (e.g., KFERQ-PA-mCherry-EGFP) | Core tool. Ensure the KFERQ motif is intact and accessible. Validate sequence. |
| High-Efficiency Transfection Reagents (e.g., Lipofectamine 3000, polyethylenimine) | Maximize reporter uptake in hard-to-transfect cells (e.g., primary neurons). |
| Lentiviral Vectors for Reporter | For stable cell line generation or infecting difficult cells, ensuring consistent expression. |
| Anti-GFP/RFP/mCherry Antibodies | For Western Blot validation of reporter expression and integrity. |
Compromised Cellular CMA Capacity
The assay measures relative activity; baseline CMA may be low due to physiological or pathological states.
Protocol: Induction of CMA as a Positive Control
- Serum Starvation: Incubate reporter-expressing cells in serum-free medium for 12-16 hours. This classic CMA inducer increases lysosomal uptake of CMA substrates.
- Pharmacological Inhibition: Treat control cells with 10 mM 3-Methyladenine (3-MA) for 6 hours (inhibits early macroautophagy, which can compete for lysosomal resources) OR use KNK437 (HSP70 inhibitor) to specifically blunt CMA.
- Quantification: Compare mCherry/EGFP ratios or mCherry-only puncta counts between induced, basal, and inhibited conditions.
Lysosomal Impairment or Altered pH
Successful CMA requires functional lysosomes with intact H+ gradients. Reporter signal depends on lysosomal acidification to quench EGFP.
Protocol: Assessment of Lysosomal Function & pH
- LysoTracker Staining: Co-stain reporter-expressing cells with 50 nM LysoTracker Green for 30 min. Image. Poor or absent staining suggests lysosomal mass/acidification defects.
- LAMP2A Co-staining: Immunofluorescence for LAMP2A, the CMA receptor. Its lysosomal localization should increase with CMA induction.
- Bafilomycin A1 Control: Treat cells with 100 nM Bafilomycin A1 (V-ATPase inhibitor) for 4-6 hours. This should block EGFP quenching, reducing the mCherry/EGFP ratio.
Inadequate Experimental Timing
CMA is a dynamic process. Imaging too early may miss substrate accumulation.
Protocol: Kinetic Analysis
- Image reporter-expressing cells at multiple time points post-transfection (e.g., 24, 48, 72 h) and post-induction (e.g., 0, 6, 12, 24 h of serum starvation).
- Plot mCherry/EGFP ratio over time to identify the peak activity window for your specific cell model.
Table 1: Impact of Common Treatments on CMA Reporter Readouts
| Experimental Condition | Expected Effect on mCherry/EGFP Ratio | Expected Effect on mCherry-Only Puncta | Purpose in Troubleshooting |
|---|---|---|---|
| Basal (Complete Medium) | Baseline (e.g., 1.0) | Low | Control for normal CMA flux. |
| Serum Starvation (12-16h) | Increase (e.g., 2.5 - 4.0) | Marked Increase | Positive control; confirms system responsiveness. |
| Bafilomycin A1 (100 nM, 4-6h) | Decrease (e.g., 0.3 - 0.6) | Decrease | Tests lysosomal acidification dependence. |
| KNK437 (HSP70 inhibitor) | Decrease | Decrease | Tests specificity of CMA blockade. |
| LAMP2A siRNA Knockdown | Decrease | Decrease | Confirms CMA-specificity of signal. |
Table 2: Troubleshooting Decision Matrix Based on Control Outcomes
| Positive Control (Starvation) Result | Bafilomycin A1 Control Result | Likely Issue | Next Step |
|---|---|---|---|
| No Increase | No Effect | Reporter defect or CMA pathway severely impaired. | Validate reporter expression via WB. Check LAMP2A levels. |
| No Increase | Decrease | Basal activity present, but induction blocked. | Check stress/induction pathways (e.g., oxidative stress). |
| Normal Increase | No Decrease | Signal may not be lysosomal/CMA-specific. | Check for artifacts; use LAMP2A co-localization. |
| Normal Increase | Normal Decrease | Assay is functional. Low basal signal is biological. | Proceed with experimental variables. Use longer induction. |
Essential Signaling Pathways & Workflows
Title: Core CMA Substrate Processing Pathway
Title: Low Signal Troubleshooting Decision Tree
Resolving low signal in CMA reporter assays requires a methodical approach that validates the reporter tool, confirms the cellular CMA machinery's competence, and verifies assay specificity. By implementing the positive and negative controls and quantitative frameworks outlined here, researchers can robustly measure CMA activity, advancing our understanding of its critical role in cellular protein quality control and its potential as a therapeutic target.
Validating Pharmacological Modulator Specificity and Off-Target Effects
Within the burgeoning field of cellular protein quality control, chaperone-mediated autophagy (CMA) has emerged as a critical, selective degradation pathway. Its dysfunction is implicated in aging, neurodegenerative diseases, and cancer, making it a prime therapeutic target. The development and application of pharmacological CMA modulators—both activators and inhibitors—is therefore of intense interest. However, the utility of any chemical probe or therapeutic candidate hinges on rigorous validation of its specificity and a comprehensive understanding of its off-target effects. This guide provides a technical framework for this essential validation process, situated within the broader research thesis that precise manipulation of CMA function is key to deciphering its physiological roles and therapeutic potential.
Key Pharmacological CMA Modulators: Current Landscape
The following table summarizes the most cited pharmacological agents affecting CMA, their intended molecular target, and known primary challenges regarding specificity.
Table 1: Common Pharmacological CMA Modulators and Specificity Considerations
| Modulator Name | Primary Intended Target/Effect on CMA | Key Reported Off-Targets/Concerns | Typical Working Concentration |
|---|---|---|---|
| CA-77.1 (Activator) | LAMP-2A stabilization; CMA activation. | Potential interaction with other lysosomal receptors; effects on macroautophagy flux. | 10-20 µM |
| Xestospongin B (Inhibitor) | Reported to block substrate translocation. | Well-established as an IP3 receptor inhibitor; affects calcium signaling broadly. | 5-10 µM |
| Bafilomycin A1 (Used to block CMA degradation) | V-ATPase inhibitor; lysosomal acidification blockade. | Inhibits all autophagic pathways (macro, micro, CMA) and endo-lysosomal trafficking. | 50-100 nM |
| 6-Aminonicotinamide (6-AN) (Activator in stress contexts) | Metabolic stress inducer, secondary CMA upregulation. | General inhibitor of glycolysis (targets G6PD); pleiotropic cellular stress. | 100-250 µM |
| Peptide Conjugates (e.g., P140) | Modulates HSPA8/HSC70 activity or substrate binding. | Potential immunomodulatory effects; specificity for CMA-related vs. other HSPA8 functions. | Varies (µM range) |
A Hierarchical Framework for Specificity Validation
Validation must move beyond single-assay confirmation. A multi-tiered approach is recommended.
Tier 1: Confirmation of On-Target Effect in CMA-Compromised Systems
The most direct test of specificity is the abolition of the modulator's effect in a system where the intended target is genetically absent or depleted.
- Experimental Protocol (Genetic Rescue/Abrogation):
- Use siRNA or CRISPR-Cas9 to knock down (KD) or knock out (KO) the core CMA component LAMP2A in the relevant cell line (e.g., mouse embryonic fibroblasts, cancer cells).
- Validate KD/KO efficiency via western blot (LAMP-2A) and functional CMA assay (e.g., fluorescence-based reporter flux).
- Treat isogenic control (scrambled siRNA or WT) and LAMP2A-KD/KO cells with the candidate modulator.
- Measure a direct CMA output. A specific CMA activator will fail to increase CMA activity in LAMP2A-deficient cells. A specific inhibitor will fail to inhibit it.
- Key Controls: Include a positive control (e.g., serum starvation for activation) and monitor cell viability (MTT/ATP assay) to rule out cytotoxicity.
Tier 2: Exclusion of Cross-Talk with Other Proteolytic Pathways
A modulator must be shown to selectively impact CMA without inadvertently altering macroautophagy or the ubiquitin-proteasome system (UPS).
- Experimental Protocol (Parallel Pathway Flux Analysis):
- CMA Flux: Employ the KFERQ-PA-mCherry-EGFP reporter. The acid-sensitive EGFP quenches in the lysosome, while mCherry is stable. CMA flux is quantified by the mCherry/EGFP signal ratio or accumulation of the cleaved reporter product via immunoblot.
- Macroautophagy Flux: Use an LC3-II turnover assay. Treat cells with modulator in the presence vs. absence of lysosomal inhibitors (Bafilomycin A1, 100 nM, or Chloroquine, 50 µM for 4-6h). Measure LC3-II accumulation by western blot. Increased flux = lower LC3-II without inhibitor, greater accumulation with inhibitor.
- UPS Activity: Utilize a ubiquitin-proteasome reporter (e.g., Ub~GFP~). Treat cells with the modulator and a proteasome inhibitor (MG132, 10 µM, 6h) as a control. Monitor GFP accumulation via fluorescence or immunoblot. Specific CMA modulators should not alter Ub~GFP~ degradation kinetics.
- Perform all three assays in parallel under identical treatment conditions. Data should be summarized in a comparative table (see Table 2).
Table 2: Hypothetical Data from Multi-Pathway Specificity Screening
| Treatment | CMA Flux (KFERQ Reporter Signal) | Macroautophagy Flux (LC3-II Turnover) | Proteasome Activity (Ub~GFP~ Degradation) | Interpretation |
|---|---|---|---|---|
| Control (DMSO) | 1.0 (Baseline) | 1.0 (Baseline) | 1.0 (Baseline) | - |
| CMA Activator (X) | 2.5 | 1.1 | 0.9 | Specific CMA activation. |
| CMA Inhibitor (Y) | 0.3 | 0.9 | 1.0 | Specific CMA inhibition. |
| Known Off-Target Agent (Z) | 1.8 | 0.2 (Inhibited) | 1.2 | Non-specific; inhibits macroautophagy. |
| Bafilomycin A1 | 0.1 | 0.1 | 1.1 | Pan-lysosomal inhibitor. |
| MG132 | 1.0 | 1.2 | 0.1 | Proteasome inhibitor. |
Tier 3: Global Profiling for Off-Target Identification
Chemical proteomics and transcriptomics/proteomics are essential for unbiased discovery.
- Experimental Protocol (Cellular Thermal Shift Assay - CETSA):
- Treat live cells or cell lysates with the modulator or vehicle.
- Heat aliquots of the sample to a range of temperatures (e.g., 37°C to 65°C).
- Separate soluble (non-denatured) protein from aggregates by centrifugation.
- Analyze the soluble fraction by quantitative mass spectrometry (MS).
- Proteins whose thermal stability shifts in the presence of the drug are direct or indirect binding targets. Compare the list of stabilized/destabilized proteins to the expected target (e.g., LAMP-2A, HSPA8). This identifies potential off-targets.
- Experimental Protocol (Phosphoproteomics/Transcriptomics):
- Treat cells with the modulator at the working concentration for a relevant time course (e.g., 2h, 8h, 24h).
- Harvest cells and perform global phosphoproteomic analysis (via LC-MS/MS) or RNA-Seq.
- Bioinformatics analysis (pathway enrichment, upstream regulator prediction) reveals which signaling networks are perturbed. Activation of stress pathways (e.g., UPR, oxidative stress) or other degradation pathways indicates compensatory or off-target effects.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for CMA Modulator Validation
| Reagent Category | Specific Item/Assay | Function in Validation |
|---|---|---|
| CMA Activity Reporters | KFERQ-PA-mCherry-EGFP (Plasmid/virus) | Direct, quantitative measurement of CMA flux in live or fixed cells. |
| Genetic Tools | LAMP2A siRNA/shRNA; LAMP2A KO cell lines (CRISPR) | Essential for Tier 1 genetic rescue experiments to prove target specificity. |
| Pathway-Specific Reporters | LC3-GFP/RFP (for macroautophagy); Ub~GFP~ (for UPS) | Parallel pathway assessment to rule out cross-talk (Tier 2). |
| Lysosomal Function Probes | LysoTracker Dyes, Magic Red Cathepsin Assay | Assess lysosomal health and general activity; modulators should not cause gross lysosomal dysfunction. |
| Global Profiling Tools | CETSA-MS Kit, Phosphoproteomics Services (e.g., TMT labeling) | Unbiased identification of direct binding targets and affected signaling pathways (Tier 3). |
| Viability Assays | Real-time ATP monitoring (e.g., CellTiter-Glo), Caspase-3/7 assay | Distinguish specific pharmacological effects from general cytotoxicity. |
Visualizing the Validation Workflow and CMA Pathway
Diagram 1: Hierarchical Modulator Validation Workflow (96 chars)
Diagram 2: CMA Pathway & Pharmacological Modulation Points (99 chars)
The path to a reliable CMA-targeting pharmacology is paved with rigorous validation. By employing a tiered strategy—from genetic target confirmation and parallel pathway screening to unbiased global profiling—researchers can confidently classify compounds as specific chemical probes or multi-target agents. This diligence is paramount for the core thesis of CMA research: only with specific tools can we accurately deconvolute CMA's role in cellular proteostasis and disease, enabling the rational development of therapeutics with predictable and on-target actions.
Within the broader thesis on chaperone-mediated autophagy (CMA) function in cellular protein quality control research, quantifying CMA activity and establishing its direct correlation with physiological and pathological outcomes is paramount. This guide details the core methodologies, data interpretation, and reagent tools essential for this endeavor.
Quantitative Measures of CMA Activity
CMA activity can be assessed through multiple, complementary quantitative readouts. The following table consolidates key metrics.
Table 1: Core Quantitative Assays for CMA Activity Measurement
| Assay Name | Measured Parameter | Typical Output/Units | Functional Correlation |
|---|---|---|---|
| LAMP2A Multimerization Assay | Percentage of LAMP2A in lysosomal membranes organized into multimeric translocation complexes. | % LAMP2A in high-MW complexes. | Directly proportional to CMA capacity. |
| KFERQ-Dendra2 Flux Assay | Rate of photoconverted CMA substrate degradation in lysosomes. | Half-life (t½) of photoconverted signal; Flux (AU/time). | Inverse correlation: shorter t½ = higher CMA activity. |
| Lyso-CIMA | Colocalization of CMA substrates with lysosomes under induced conditions. | Colocalization coefficient (Manders' M1/M2). | Direct correlation: higher coefficient = higher CMA activity. |
| CMA Reporter Cell Lines (e.g., CMA-Rosella) | Lysosomal delivery and quenching of pH-sensitive reporter. | Ratio of lysosomal (quenched) to cytosolic signal. | Direct correlation: higher ratio = higher CMA activity. |
Detailed Experimental Protocols
1. Protocol: LAMP2A Multimerization by Blue Native-PAGE
- Principle: Resolve native LAMP2A complexes from lysosomal membranes to assess the active translocation complex formation.
- Methodology:
- Lysosome Isolation: Purify lysosomes from tissue/cells using discontinuous Percoll density gradient centrifugation.
- Membrane Solubilization: Solubilize lysosomal pellets in ice-cold 1% Digitonin in Native-PAGE buffer for 30 min.
- Electrophoresis: Centrifuge solubilized material, load supernatant onto 4-16% Bis-Tris Blue Native-PAGE gel. Run at 4°C, 100V.
- Detection: Immunoblot for LAMP2A. Monomeric LAMP2A runs at ~96 kDa; functional multimeric complexes appear as high-molecular-weight bands (>700 kDa).
- Data Interpretation: The ratio of multimeric to total LAMP2A signal quantifies CMA capacity.
2. Protocol: KFERQ-Dendra2 Photoconversion Flux Assay
- Principle: A photo-switchable CMA substrate allows precise temporal measurement of lysosomal degradation.
- Methodology:
- Transfection: Express KFERQ-tagged Dendra2 in cells.
- Photoconversion: Use a 405nm laser to convert a region-of-interest from green to red fluorescence.
- Time-Lapse Imaging: Monitor the decay of red (photoconverted) signal and the steady-state green signal over 6-24 hours using live-cell confocal microscopy.
- Inhibition Control: Co-treat with lysosomal inhibitors (e.g., E64d/Pepstatin A) to confirm lysosomal degradation.
- Data Interpretation: Fit the red fluorescence decay curve to a one-phase decay model. The half-life (t½) is the primary metric. Shorter t½ indicates higher CMA flux.
Visualizing the CMA Pathway & Experimental Workflow
Diagram 1: CMA Substrate Translocation Pathway
Diagram 2: KFERQ-Dendra2 Flux Assay Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CMA Activity Research
| Reagent / Material | Function & Application |
|---|---|
| Anti-LAMP2A (clone EPR17713 / ab18528) | Specific antibody for detecting the CMA-specific splice variant of LAMP2 via immunoblot, immunofluorescence, or immunoprecipitation. |
| KFERQ-Dendra2 Plasmid | Photo-switchable CMA reporter construct for live-cell measurement of CMA substrate flux. |
| Lysosome Isolation Kit (e.g., from Sigma) | Kit for rapid purification of intact lysosomes from tissues or cultured cells for biochemical assays. |
| Digitonin (High-Purity) | Mild detergent for solubilizing lysosomal membranes while preserving native protein complexes for Blue Native-PAGE. |
| CMA Inhibitor (e.g., P140 peptide) | Experimental tool to acutely inhibit substrate binding to HSC70, used as a negative control. |
| Lysosomal Protease Inhibitors (E64d & Pepstatin A) | Cocktail used to inhibit lysosomal degradation, confirming that signal loss is due to proteolysis. |
| Recombinant HSC70 Protein | For in vitro validation of substrate binding via KFERQ motif interaction studies. |
| CMA Reporter Cell Line (e.g., CMA-Rosella) | Stable cell line expressing a ratiometric pH-sensitive CMA reporter for high-throughput screening. |
CMA in Context: Comparative Analysis with UPS and Macroautophagy in Proteostasis
Within the eukaryotic proteostasis network, the degradation of intracellular proteins is primarily governed by three systems: the Ubiquitin-Proteasome System (UPS), macroautophagy, and Chaperone-Mediated Autophagy (CMA). While the UPS and macroautophagy have been extensively characterized, CMA represents a more selective and mechanistically distinct pathway whose full functional integration is still being elucidated. The central thesis of modern proteostasis research posits that CMA is not merely a redundant backup but a critical, complementary regulator with unique substrate specificity, activation kinetics, and roles in cellular stress adaptation. Its dysfunction is increasingly linked to aging, neurodegenerative diseases, and metabolic disorders, making it a compelling target for therapeutic intervention. This guide provides a technical dissection of these three systems, emphasizing their synergistic and non-overlapping functions.
Core Mechanisms and Comparative Analysis
Chaperone-Mediated Autophagy (CMA)
CMA directly translocates individual cytosolic proteins across the lysosomal membrane. Substrate recognition is initiated by the chaperone HSC70, which identifies a pentapeptide motif (KFERQ-like) in target proteins. The substrate-chaperone complex docks at the lysosomal membrane via interaction with the single-span receptor protein LAMP2A. Monomeric LAMP2A multimerizes to form a translocation complex, a rate-limiting step regulated by a luminal subcomplex (GFAP, EF1α). The substrate is unfolded and translocated into the lumen with the assistance of a luminal isoform of HSC70, where it is rapidly degraded.
Ubiquitin-Proteasome System (UPS)
The UPS targets primarily short-lived, misfolded, or regulatory proteins for degradation. It involves a cascade: ubiquitin activation (E1), conjugation (E2), and ligation (E3) to the substrate, often forming polyubiquitin chains (typically Lys48-linked). The tagged substrate is recognized and degraded by the 26S proteasome, a multiprotease complex, yielding short peptides. This process is ATP-dependent and essential for rapid signaling modulation and the clearance of soluble misfolded proteins.
Macroautophagy
Macroautophagy engulfs large cytoplasmic cargoes—protein aggregates, damaged organelles, or pathogens—within a double-membraned vesicle, the autophagosome. Initiation involves the ULK1/ATG1 complex and the PI3K class III complex, leading to phagophore nucleation. Cargo can be sequestered non-selectively or via selective receptors (e.g., p62/SQSTM1, NBR1) that bind ubiquitinylated cargo and LC3 on the expanding phagophore. The completed autophagosome fuses with a lysosome, forming an autolysosome where contents are degraded.
Quantitative Comparison of Degradation Pathways
Table 1: Core Characteristics of the Major Proteolytic Systems
| Feature | Chaperone-Mediated Autophagy (CMA) | Ubiquitin-Proteasome System (UPS) | Macroautophagy |
|---|---|---|---|
| Degradation Cargo | Soluble proteins with KFERQ-like motif | Short-lived & misfolded soluble proteins, polyubiquitinated | Bulk cytoplasm, aggregates, organelles (selective/non-selective) |
| Selectivity | High (motif-dependent) | High (ubiquitin-tag dependent) | Can be non-selective or receptor-mediated selective |
| Degradation Rate | Intermediate (hours) | Very Rapid (minutes) | Slow (hours) |
| Cargo Translocation | Direct across lysosomal membrane (unfolded) | Through proteasome core (unfolded) | Vesicular sequestration |
| Key Receptor | LAMP2A | Proteasome (Rpn10/S5a) | Autophagy receptors (p62, NBR1, OPTN) |
| Key Regulatory Step | LAMP2A multimerization | Polyubiquitination by E3 ligases | Phagophore elongation (LC3 lipidation) |
| Primary Energy Source | ATP (for chaperone function) | ATP (unfolding, proteasome activity) | ATP (membrane formation, fusion) |
| Response to Stress | Activated by oxidative stress, nutrient depletion | Inhibited by severe oxidative stress | Activated by nutrient depletion, proteotoxic stress |
Table 2: Physiological and Pathological Roles
| Context | CMA Role | UPS Role | Macroautophagy Role |
|---|---|---|---|
| Nutrient Starvation | Provides amino acids via selective degradation of specific proteins. | Activity generally decreases due to reduced ATP. | Induced for bulk recycling of cytoplasmic components. |
| Oxidative Stress | Degrades oxidized proteins to prevent aggregation; upregulated. | Can be impaired if proteasome subunits are damaged. | Removes damaged mitochondria (mitophagy) and protein aggregates. |
| Aging | Activity declines due to reduced LAMP2A levels; contributes to proteostasis collapse. | Activity and efficiency generally decline. | Basal activity declines; implicated in longevity. |
| Neurodegeneration (e.g., PD, AD) | Impaired; α-synuclein and tau are CMA substrates that can block the pathway when mutated/aggregated. | Impaired; aggregation-prone proteins often inhibit proteasome function. | Often impaired; crucial for clearing protein aggregates and damaged mitochondria. |
| Cancer | Can be pro- or anti-tumorigenic; supports tumor cell survival under metabolic stress. | Highly active; degrades tumor suppressors; often targeted by proteasome inhibitors. | Can be pro- or anti-tumorigenic; supports survival or induces cell death. |
Experimental Methodologies
Assessing CMA Activity
Protocol: Lysosomal Binding and Uptake Assay
- Isolation of Lysosome-Rich Fractions: Homogenize rat liver or cultured cells in ice-cold 0.25 M sucrose buffer. Perform differential centrifugation (800 x g, 10 min; 10,000 x g, 20 min; final supernatant at 100,000 x g, 60 min). The pellet is the light mitochondrial-lysosomal fraction.
- Preparation of CMA Substrate: Isolate GAPDH (a known CMA substrate) or use recombinant protein. Radiolabel with 125I or conjugate to a fluorescent dye (e.g., Cy5).
- Binding/Uptake Assay: Incubate lysosomal fraction (50-100 μg protein) with substrate (5-10 nM) in uptake buffer (10 mM HEPES, 0.3 M sucrose, 1 mM DTT, 5 mM MgCl2, pH 7.4) ± ATP-regenerating system and protease inhibitors (to measure uptake vs. binding/degradation). For binding-only assays, include 0.1% Triton X-100 to disrupt membranes.
- Separation and Quantification: After incubation (20-40 min, 37°C), separate lysosomes by centrifugation through a 4% BSA cushion. Measure radioactivity/fluorescence in the lysosomal pellet. Uptake is calculated as the protease-protected signal.
Measuring UPS Activity
Protocol: Proteasome Chymotrypsin-like Activity Assay
- Cell Lysate Preparation: Lyse cells in hypotonic buffer (50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM DTT, 0.5 mM EDTA, 10% glycerol) followed by centrifugation (20,000 x g, 15 min).
- Reaction Setup: In a 96-well plate, mix 20-50 μg of lysate protein with assay buffer (50 mM Tris-HCl, pH 7.5, 5 mM MgCl2, 1 mM DTT) containing 100 μM fluorogenic substrate (Suc-LLVY-AMC for chymotrypsin-like activity). The AMC group fluoresces upon cleavage.
- Inhibition Control: Include a well with the specific proteasome inhibitor MG-132 (20 μM) to confirm signal specificity.
- Kinetic Measurement: Immediately monitor fluorescence (Ex 380 nm/Em 460 nm) in a plate reader at 37°C for 30-60 minutes. Activity is expressed as the rate of AMC release (RFU/min) normalized to protein content.
Monitoring Macroautophagy Flux
Protocol: LC3-II Turnover Assay via Immunoblot
- Treatment: Treat cells to induce autophagy (e.g., starvation in EBSS medium, treatment with mTOR inhibitor like Torin1). To block lysosomal degradation, include an inhibitor like Bafilomycin A1 (100 nM) or Chloroquine (50 μM) for the last 2-4 hours of treatment.
- Sample Collection: Harvest cells in direct Laemmli sample buffer to prevent protein degradation.
- Immunoblotting: Perform SDS-PAGE and western blot for LC3. Detect both the cytosolic form (LC3-I, ~16 kDa) and the lipidated, autophagosome-associated form (LC3-II, ~14 kDa).
- Interpretation: An increase in LC3-II levels in the presence of lysosomal inhibitors (compared to without inhibitors) indicates increased autophagic flux (i.e., increased delivery to lysosomes). A static LC3-II level without inhibitors is difficult to interpret.
Diagrammatic Representations
Diagram 1: CMA Substrate Translocation Pathway
Diagram 2: Macroautophagy Induction and Flux
Diagram 3: Proteostasis Network Decision Logic Under Stress
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Key Reagents for Studying Proteostasis Pathways
| Reagent | Target/Function | Application | Key Provider Examples |
|---|---|---|---|
| MG-132 / Bortezomib | 26S Proteasome inhibitor (blocks chymotrypsin-like activity). | Inhibiting UPS function to assess compensatory pathways or protein stabilization. | Sigma-Aldrich, Selleckchem, MedChemExpress |
| Bafilomycin A1 / Chloroquine | Vacuolar-type H+-ATPase inhibitor / Lysosomotropic agent; blocks autophagic flux. | Measuring autophagic flux (LC3-II turnover) and distinguishing induction from degradation block. | Cayman Chemical, Tocris, Sigma-Aldrich |
| Torin 1 / Rapamycin | mTOR kinase inhibitor / mTORC1 inhibitor; potent inducer of macroautophagy. | Experimentally inducing autophagy under nutrient-rich conditions. | Tocris, Cell Signaling Tech, Sigma-Aldrich |
| Anti-LAMP2A antibody (clone EPR8475) | Specifically recognizes the CMA-specific isoform LAMP2A. | Measuring LAMP2A levels by immunoblot or immunofluorescence; key for CMA capacity. | Abcam |
| Anti-LC3B antibody | Detects both LC3-I and LC3-II forms. | Gold standard for monitoring autophagosome formation and flux via immunoblot/IF. | Cell Signaling Tech, Novus, MBL |
| Anti-p62/SQSTM1 antibody | Detects the autophagy receptor and ubiquitin-binding protein. | Assessing selective autophagy and autophagic flux (p62 degradation). | Cell Signaling Tech, Abnova, MBL |
| Cy5-labeled GAPDH | Fluorescently labeled canonical CMA substrate. | In vitro assays for measuring lysosomal binding and uptake in isolated fractions. | Custom conjugation or available from Protea Biosciences |
| Puromycin-linker constructs | Generate puromycin-conjugated proteins in cells via expression of a puromycin N-terminal linker. | Measuring global protein degradation rates (e.g., in DRIP, D2P assays). | Available upon request from relevant labs (Goldberg, Kirchner et al.) |
| HSPA8/HSC70 Activators (e.g., YM-1) | Allosteric activator of HSC70 ATPase activity. | Experimentally enhancing CMA activity in cellular or animal models. | Under development; research-grade from academic labs. |
| LAMP2A shRNA/CRISPR | Genetic knock-down or knock-out of the CMA receptor. | Validating CMA-specific phenotypes and confirming substrate reliance on CMA. | Santa Cruz, Horizon, Synthego |
Chaperone-Mediated Autophagy (CMA) is a selective lysosomal degradation pathway integral to cellular proteostasis. Within the broader thesis of CMA's role in cellular protein quality control, three core pillars define its functionality and regulatory complexity: Selectivity (the precision in substrate recognition), Capacity (the maximal throughput of the pathway), and Physiological Triggers (the signals that modulate CMA activity). Understanding the interplay between these elements is critical for elucidating CMA's contribution to health, aging, and disease, and for informing therapeutic strategies in neurodegeneration, cancer, and metabolic disorders.
Selectivity: The Molecular Basis of Substrate Recognition
CMA selectivity is conferred by a pentapeptide motif (KFERQ-like) in substrate proteins. Recognition is mediated by the cytosolic chaperone Hsc70 (HSPA8) and its co-chaperones, which target the substrate to the lysosomal membrane receptor LAMP2A.
Key Experimental Protocol: Identifying CMA Substrates
Method: In vitro CMA assay using isolated lysosomes.
- Lysosome Isolation: Purify lysosomes from rat liver or cultured cells via centrifugation on a discontinuous metrizamide density gradient.
- Substrate Preparation: Radiolabel (e.g., ¹⁴C) or fluorescently tag candidate proteins via in vitro translation or chemical conjugation.
- Binding/Uptake Assay: Incubate purified lysosomes with the substrate in CMA reaction buffer (e.g., 10 mM HEPES, 0.3 M sucrose, 1 mM DTT, 5 mM MgCl₂, pH 7.4) at 37°C.
- Protease Protection: After incubation, treat with proteinase K to degrade externally bound, non-internalized substrate.
- Analysis: Resolve lysosomal proteins by SDS-PAGE and quantify substrate uptake via autoradiography or fluorescence imaging. Specificity is confirmed by competition with a known CMA substrate (e.g., GAPDH) or using lysosomes from LAMP2A-deficient models.
Quantitative Data: Selectivity Parameters
Table 1: Characteristics of Canonical CMA Substrates
| Substrate Protein | KFERQ-like Motif | Cellular Function | Half-life Reduction with CMA Activation |
|---|---|---|---|
| GAPDH | QDKFH | Glycolysis | ~40% |
| RNase A | KFERQ | RNA digestion | ~60% |
| IκBα | EDLQR | NF-κB signaling | ~50% |
| MEF2D | QKIVE | Transcription | ~55% |
| α-Synuclein | VKKDQ | Neuronal function | ~70% |
Capacity: Regulation of Lysosomal Uptake and Throughput
CMA capacity is determined by the levels and assembly state of LAMP2A at the lysosomal membrane. Multimeric LAMP2A complexes form the active translocation structure. Capacity is dynamically regulated by rates of lysosomal membrane insertion, stabilization, and cleavage.
Key Experimental Protocol: Measuring CMA Activity Flux
Method: Photoactivatable Fluorescent Reporter Assay (KFERQ-PA-mCherry).
- Reporter Construction: Stably transduce cells with a construct expressing a CMA substrate motif (KFERQ) fused to photoactivatable mCherry and a constitutively expressed GFP (transfection control).
- Photoactivation & Chase: Activate mCherry in a defined region of the cytoplasm using 405 nm laser. Monitor fluorescence over 4-16 hours.
- Lysosomal Inhibition Control: Parallel experiments with lysosomal inhibitors (e.g., Bafilomycin A1, 20 nM) confirm CMA-dependent degradation.
- Quantification: Measure the decay rate of photoactivated red fluorescence normalized to the stable GFP signal using live-cell imaging and kinetic analysis software.
Quantitative Data: Capacity Metrics
Table 2: Factors Modulating CMA Capacity
| Regulatory Factor | Effect on LAMP2A | Net Effect on CMA Capacity | Experimental Change |
|---|---|---|---|
| Lysosomal Cholesterol | Stabilizes multimer | Increases (up to 3-fold) | Depletion reduces uptake by ~70% |
| GFAP / EF1α | Binds to luminal tail; regulates disassembly | Modulates (inhibits) | GFAP knockout increases uptake 2-fold |
| RAC1 / RHOA GTPases | Modulate actin dynamics at lysosome | RAC1 increases; RHOA decreases | RAC1 activation: +150%; RHOA activation: -60% |
| Oxidative Stress (H₂O₂) | Upregulates LAMP2 transcription | Increases (2-4 fold) | 200 µM H₂O₂ induces maximal response |
| Aging | Increased lysosomal instability | Decreases (up to 70% loss) | Old vs. young rodent models |
Physiological Triggers: Signaling to the CMA Machinery
CMA is activated by diverse stimuli including nutrient deprivation, oxidative stress, hypoxia, and proteotoxic stress. Key signaling hubs involve the transcription factor TFEB, reactive oxygen species (ROS), and energy-sensing kinases.
Diagram 1: Major Signaling Pathways Regulating CMA
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for CMA Research
| Reagent / Tool | Function/Application | Key Provider Examples |
|---|---|---|
| Anti-LAMP2A (clone EPR8470) | Specific detection of LAMP2A isoform via WB, IF | Abcam, Sigma-Aldrich |
| CMA Reporter (KFERQ-PA-mCherry) | Live-cell, quantitative flux measurement | Addgene (ptfLC3), custom synthesis |
| Lysosome Isolation Kit | Purification of intact lysosomes for in vitro assays | Sigma (LYSISO1), Thermo Scientific |
| Recombinant Hsc70 (HSPA8) Protein | For substrate binding and translocation studies | Enzo Life Sciences, Novus Biologicals |
| Bafilomycin A1 | V-ATPase inhibitor; blocks lysosomal acidification & degradation | Cayman Chemical, Tocris |
| LAMP2A siRNA/shRNA | Knockdown to confirm CMA-specific effects | Dharmacon, Santa Cruz Biotechnology |
| Anti-KCMA Substrate Antibody | Detects endogenous CMA substrates pre/post degradation | Custom development |
| CMA Activator (e.g., CA77.1) | Small molecule enhancer of LAMP2A expression | Under research, not commercially available |
Diagram 2: Core CMA Experimental Workflow
Synthesis and Future Perspectives
The functional output of CMA is determined by the dynamic intersection of its Selectivity (dictated by substrate motifs and chaperone availability), Capacity (governed by LAMP2A dynamics), and response to Physiological Triggers. Current drug development efforts focus on modulating this intersection—e.g., small molecules that stabilize LAMP2A multimers to boost capacity in aging or neurodegenerative contexts. Future research must employ the integrated experimental approaches outlined here to map the precise signaling networks that recalibrate selectivity under stress and to quantify capacity limits in disease models, thereby validating CMA as a therapeutic target for proteostasis-linked pathologies.
Cellular protein quality control (PQC) systems, including the ubiquitin-proteasome system (UPS), macroautophagy, and chaperone-mediated autophagy (CMA), function in a coordinated network to maintain proteostasis. CMA is a selective lysosomal degradation pathway for cytosolic proteins containing a KFERQ-like motif. Its dysfunction is linked to neurodegenerative diseases, cancer, and aging. A critical, yet incompletely resolved, concept in PQC is the phenomenon of cross-talk and compensation between these degradation pathways. When one pathway is compromised—either genetically or pharmacologically—others can be upregulated or rerouted to handle the accumulating substrate load, masking the full phenotypic impact of the initial defect. This whitepaper synthesizes current evidence from genetic and pharmacological studies that illuminate this complex interplay, with a specific focus on implications for understanding CMA function and for therapeutic targeting.
Evidence from Genetic Studies
Genetic ablation of core CMA components, such as LAMP2A (the lysosomal receptor) or HSC70 (the cytosolic chaperone), provides foundational evidence for compensatory mechanisms.
Key Observations:
- Upregulation of Macroautophagy: In LAMP2A knockout mouse liver and cultured cells, there is a consistent increase in markers of macroautophagy, such as LC3-II lipidation and numbers of autophagosomes.
- Proteasomal Activation: In some cell types, inhibition of CMA leads to an increase in polyubiquitinated proteins and proteasome activity, suggesting a shift toward UPS-mediated degradation.
- Disease Model Insights: In neurodegenerative disease models (e.g., expressing mutant α-synuclein), concomitant knockout of CMA and macroautophagy genes (e.g., Atg5 or Atg7) exacerbates pathology far more severely than impairment of either pathway alone, indicating functional compensation in vivo.
Table 1: Compensatory Responses to Genetic Inhibition of CMA
| Genetic Model | Primary Defect | Observed Compensatory Upregulation | Quantitative Measure (Typical Fold Change vs. WT) | Functional Consequence |
|---|---|---|---|---|
| Liver-specific LAMP2A KO Mouse | Loss of CMA substrate translocation | Macroautophagy flux | LC3-II/I ratio: ~2.5x; p62 degradation: +40% | Partial maintenance of protein degradation capacity |
| Fibroblast LAMP2A KO | Accumulation of KFERQ proteins | Ubiquitin-Proteasome System (UPS) activity | 20S proteasome activity: ~1.8x; Ubiquitin conjugates: +60% | Altered degradation preference, potential ER stress |
| Neuronal CMA Impairment | Alphasynuclein accumulation | Macroautophagy initiation | ATG5/7 expression: ~1.5-2x; Autophagosome count: +300% | Delayed onset of aggregation phenotypes |
Evidence from Pharmacological Studies
Pharmacological tools allow acute, tunable inhibition of specific pathways, revealing dynamic and immediate compensatory cross-talk.
Key Observations:
- Acute CMA Inhibition: Compounds like P140 (a peptide inhibiting HSC70 binding to LAMP2A) or XPO1 inhibitors (which block nuclear export of CMA transcription factors) rapidly reduce CMA flux, leading to a swift increase in macroautophagy markers within hours.
- Reciprocal Studies: Inhibition of the proteasome (e.g., with Bortezomib) or macroautophagy (e.g., with Lys05 or Chloroquine) often results in increased CMA activity, measured via lysosomal translocation of reporter proteins like KFERQ-Dendra.
- Therapeutic Implications: In cancer, simultaneous pharmacological blockade of CMA and macroautophagy induces synergistic cytotoxicity, whereas blocking only one pathway may be compensated by the other, leading to treatment resistance.
Table 2: Pharmacological Modulation Demonstrating Pathway Cross-Talk
| Pharmacological Agent | Primary Target | Concentration Range | Effect on CMA | Compensatory Response in Other Pathways |
|---|---|---|---|---|
| P140 Peptide | HSC70-LAMP2A interaction | 10-50 µM | ↓ Flux by 70-90% | Macroautophagy ↑ (LC3-II: 2-3x in 6h) |
| Bortezomib | 26S Proteasome | 10-100 nM | ↑ Activity by 50-200% | CMA as compensatory clearance route |
| Chloroquine / Lys05 | Lysosomal acidification (blocks autophagic degradation) | 10-50 µM | ↑ Activity by 40-80% | Increased KFERQ substrate binding to LAMP2A |
| Ver-155008 | HSC70 ATPase activity | 5-20 µM | ↓ Flux by ~80% | Strong upregulation of UPS and macroautophagy |
Experimental Protocols for Studying Cross-Talk
Protocol 1: Measuring CMA Flux with Compensatory Pathway Blockade
Aim: To assess the maximal degradation capacity and dependency of CMA when other pathways are inhibited.
- Cell Treatment: Seed primary mouse embryonic fibroblasts (MEFs) or relevant cell line in 6-well plates.
- Inhibition: Pre-treat cells for 4 hours with:
- DMSO (control)
- Bortezomib (100 nM) to inhibit proteasome.
- Lys05 (20 µM) to inhibit lysosomal degradation (blocks macroautophagy and CMA).
- Combination of Bortezomib + Lys05.
- CMA Activation & Measurement: Serum-starve cells (24h) to maximally induce CMA. Use the photoconvertible CMA reporter KFERQ-Dendra2.
- Photoconvert cytosolic Dendra2 from green to red.
- Chase for 4-16 hours in the continued presence of inhibitors.
- Quantify loss of red fluorescent signal (lysosomal degradation) via flow cytometry or fluorescence microscopy. Normalize to DMSO control.
- Parallel Assessment: Analyze lysates via Western blot for LC3-II, p62, and polyubiquitin chains to confirm inhibitor efficacy and compensatory shifts.
Protocol 2: Genetic Co-Inhibition in vivo
Aim: To dissect compensatory roles of macroautophagy in CMA-deficient animals.
- Model Generation: Cross LAMP2A KO mice with ATG7 floxed mice.
- Tissue-Specific Knockout: Administer AAV-Cre or use cell-type specific Cre drivers (e.g., Nestin-Cre for neurons) to generate double knockout (DKO: LAMP2A⁻/⁻; ATG7⁻/⁻) in the target tissue.
- Phenotypic Analysis: Compare at 3-12 months to single KOs and wild-type littermates.
- Biochemical: Immunoblot for disease-relevant aggregates (e.g., α-synuclein, Tau), ubiquitin, p62, and chaperones.
- Histological: Assess neurodegeneration (IHC), lipid accumulation (Oil Red O), or other organ-specific pathologies.
- Functional: Perform behavioral tests (rotarod, open field) or metabolic assays.
Visualization of Pathway Relationships and Experimental Workflows
Diagram Title: Cross-Talk and Compensation Between Protein Degradation Pathways
Diagram Title: Experimental Workflow for Measuring CMA Flux Under Compensation
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Studying CMA and Cross-Talk
| Reagent / Tool | Category | Function in Experiments | Example Product / Identifier |
|---|---|---|---|
| LAMP2A Knockout Mice/Cells | Genetic Model | Provides a system with constitutive CMA deficiency to study chronic adaptation and compensation. | Jackson Labs: B6;129-Lamp2tm1 |
| KFERQ-Dendra2 Reporter | Live-Cell Imaging Probe | A photoconvertible fluorescent protein fused to a CMA-targeting motif. Allows quantitative tracking of CMA substrate uptake and degradation over time. | Addgene #140993 (hLYZ-Dendra2) |
| P140 Peptide | Pharmacological Inhibitor | A cell-permeable peptide that binds HSC70, blocking its interaction with LAMP2A, leading to acute, specific CMA inhibition. | Custom synthesis (sequence: SGSG-GGG-[D]PRKLP-[D]NSG-[D]PRKLP) |
| Ver-155008 | Small Molecule Inhibitor | An ATP-competitive inhibitor of HSC70, inhibiting its chaperone activity. Broadly affects HSC70 functions, including CMA. | Tocris #3803 |
| Bafilomycin A1 / Lys05 | Lysosomal Inhibitor | Blocks lysosomal acidification and degradation, used to measure autophagic flux (macro and CMA) by preventing substrate breakdown. | Sigma B1793; Selleckchem S8383 |
| Anti-LAMP2A (H4B4) Antibody | Antibody | Specifically detects the LAMP2A splice variant (not 2B or 2C) by Western blot or immunofluorescence, crucial for assessing CMA capacity. | Abcam ab18528 / DSHB H4B4 |
| CMA Activity Reporter Cell Line | Stable Cell Line | Cells stably expressing a CMA reporter (e.g., KFERQ-PA-mCherry1). Provides a consistent system for high-throughput screening of modulators. | Available through core facilities (e.g., GFP-LC3-RFP-LC3ΔG modified). |
Thesis Context: Within the broader framework of cellular protein quality control, chaperone-mediated autophagy (CMA) represents a selective, lysosome-dependent degradation pathway for cytosolic proteins containing a KFERQ-like motif. This whitepaper examines the dichotomous role of CMA dysfunction in the pathogenesis of neurodegenerative diseases versus its co-option in cancer, framing this duality as central to understanding context-dependent proteostatic adaptation and failure.
CMA involves the recognition of substrate proteins by the cytosolic chaperone HSC70, delivery to the lysosomal membrane via interaction with lysosome-associated membrane protein type 2A (LAMP2A), substrate unfolding, and translocation into the lysosomal lumen for degradation. The single-span membrane protein LAMP2A is the rate-limiting component, with its levels dictating CMA activity.
Table 1: Comparative CMA Activity and Molecular Markers in Neurodegeneration vs. Cancer
| Parameter | Neurodegenerative Disease (e.g., Parkinson's, Alzheimer's) | Cancer (e.g., Pancreatic, Melanoma, Lung) | Measurement Methods |
|---|---|---|---|
| CMA Activity | Chronically reduced | Upregulated in early stages; often downregulated in advanced stages | Lysosomal uptake/ degradation assays (e.g., KFERQ-Dendra2 reporter), LAMP2A levels. |
| LAMP2A Levels | Decreased in vulnerable brain regions (e.g., ~40-60% reduction in substantia nigra in PD). | Highly variable; often elevated in tumors vs. adjacent tissue (e.g., 2-5 fold increase in some carcinomas). | Immunoblot, immunohistochemistry, transcriptomics. |
| Primary Consequence | Accumulation of toxic, aggregation-prone proteins (α-synuclein, Tau). | Enhanced stress adaptation, metabolic reprogramming, treatment resistance. | Protein aggregation assays, survival assays under stress. |
| Key Pathogenic Substrates | α-synuclein, Tau, MEF2D, UCH-L1, LRRK2. | GAPDH, PKM2, HIF1α, c-MYC, RIPK1/2. | Co-immunoprecipitation with LAMP2A/HSC70, CMA motif prediction. |
| Therapeutic Targeting Strategy | CMA activation (e.g., LAMP2A stabilizers, HSF1 activators). | CMA inhibition (e.g., LAMP2A blockers, disruptors of lysosomal uptake). | High-throughput screening for modulators, in vivo xenograft models. |
Table 2: Experimental Models and Phenotypic Outcomes
| Disease Context | Genetic Model | Phenotype | Key Readout |
|---|---|---|---|
| Neurodegeneration | Neuron-specific LAMP2A KO mice. | Accumulation of ubiquitinated proteins, motor deficits, neuronal loss. | Rotarod performance, neuron count, protein aggregates (IHC). |
| Cancer | LAMP2A shRNA knockdown in cancer cell lines. | Increased sensitivity to oxidative stress & chemotherapeutics, reduced tumor growth in vivo. | Colony formation assay, tumor volume in mice, apoptosis markers. |
| Cancer | LAMP2A overexpression in primary cells. | Immortalization, anchorage-independent growth. | Soft agar assay, proliferation rate. |
Detailed Experimental Protocols
Protocol 1: Measuring CMA Activity Using the KFERQ-Dendra2 Reporter
- Principle: A photoconvertible fluorescent protein (Dendra2) fused to a canonical CMA motif (KFERQ) is expressed in cells. Photoconversion from green to red fluorescence creates a time-stamped pool. The loss of red signal over time indicates lysosomal degradation via CMA.
- Procedure:
- Transfection: Transfect cells with plasmid encoding CMA reporter (e.g., pSelect-KFERQ-Dendra2).
- Photoconversion: At 48h post-transfection, expose cells to 405 nm light (or specific Dendra2 photoconversion setting) for 2-5 minutes to convert a subset of Dendra2 from green to red.
- Chase & Inhibition: Replace medium. For controls, include lysosomal inhibitors (e.g., 20 mM NH₄Cl + 100 µM Leupeptin) or CMA-specific inhibitor (e.g., P140 peptide targeting HSC70).
- Imaging & Quantification: Acquire red channel images immediately (T=0) and at designated chase periods (e.g., 4h, 8h, 16h). Use image analysis software to quantify total red fluorescence per cell, normalized to T=0.
- Analysis: The rate of red fluorescence loss in control vs. inhibited cells reflects CMA flux.
Protocol 2: Assessing LAMP2A Complex Assembly at the Lysosomal Membrane
- Principle: CMA activation requires multimerization of LAMP2A into a translocation complex. This can be assessed by lysosomal membrane isolation and blue native-PAGE.
- Procedure:
- Lysosome Enrichment: Homogenize tissues/cells in isotonic buffer (0.25 M sucrose, 10 mM HEPES). Perform differential centrifugation to obtain a heavy mitochondrial/lysosomal pellet.
- Membrane Solubilization: Solubilize the pellet in 1% digitonin-containing buffer (preserves protein complexes).
- Blue Native-PAGE: Load solubilized lysosomal proteins onto a native polyacrylamide gradient gel. Electrophorese under non-denaturing conditions.
- Immunoblotting: Transfer proteins to PVDF membrane and probe with anti-LAMP2A antibody.
- Analysis: LAMP2A monomers (~96 kDa), intermediate multimers (~200-400 kDa), and the high-molecular-weight translocation complex (>700 kDa) can be visualized. The ratio of complex to monomer indicates CMA capacity.
Visualization: Pathways and Workflows
Title: CMA Substrate Translocation Mechanism
Title: Divergent Disease Outcomes from CMA Dysfunction
The Scientist's Toolkit: Key Research Reagents
| Reagent / Material | Function / Application | Example Product/Source |
|---|---|---|
| Anti-LAMP2A (Clone EPR6850) | Specific antibody for detecting LAMP2A protein (not other LAMP2 isoforms) by immunoblot, IHC, or IP. | Abcam (ab18528) |
| KFERQ-Dendra2 Reporter Plasmid | Live-cell, quantitative measurement of CMA flux via photoconversion and chase assays. | Addgene (Plasmid #110067) |
| P140 Peptide | A 21-mer peptide derived from U1-70K snRNP protein; selectively inhibits CMA by blocking substrate binding to HSC70. | Custom synthesis (Sequence: GGRGDYROKPPRGVKVVIPPRR) |
| Lysosomal Inhibitor Cocktail | Inhibits lysosomal proteolysis to establish baseline in degradation assays. | 20 mM NH₄Cl + 100 µM Leupeptin |
| Digitonin | Mild detergent for solubilizing lysosomal membranes while preserving native protein complexes for BN-PAGE. | MilliporeSigma (D141) |
| Photoactivatable Cell Line | Stable cell line expressing KFERQ-PA-mCherry-EGFP for CMA tracking without transfection. | Generated via lentiviral transduction of reporter construct. |
| hsc70 (HSPA8) siRNA | Gene knockdown to validate CMA-specific substrate degradation vs. other autophagy pathways. | Dharmacon ON-TARGETplus SMARTpool |
1. Introduction: CMA in Cellular Protein Quality Control Chaperone-mediated autophagy (CMA) is a selective lysosomal degradation pathway critical for maintaining cellular proteostasis. Unlike macroautophagy, CMA directly targets individual cytosolic proteins bearing a KFERQ-like motif for translocation across the lysosomal membrane via the receptor lysosomal-associated membrane protein type 2A (LAMP2A). Within the broader protein quality control network, CMA complements the ubiquitin-proteasome system and other autophagic pathways, specifically regulating metabolic enzymes, transcription factors, and proteins implicated in neurodegeneration. Consequently, CMA dysfunction is a hallmark of aging and various pathologies, positioning LAMP2A as a pivotal biomarker for cellular health and disease.
2. Quantitative Data on LAMP2A in Aging and Disease
Table 1: LAMP2A Levels and CMA Activity in Aging Models
| Model System | LAMP2A Protein Level (vs. Young) | CMA Activity (vs. Young) | Key Measurement Method | Reference (Example) |
|---|---|---|---|---|
| Mouse Liver (24 mo) | ~30-50% decrease | ~70% decrease | Immunoblot, Photoactivatable CMA reporter | (Cuervo & Dice, 2000) |
| Rat Liver (24 mo) | ~60% decrease | ~65% decrease | Immunoblot, Lysosomal uptake assay | (Kiffin et al., 2007) |
| Human Fibroblasts (>65 yrs) | ~40% decrease | ~60-70% decrease | qPCR, Immunoblot, HsCMA reporter | (Zhang & Cuervo, 2008) |
| Mouse Brain (Hippocampus, 20 mo) | ~40% decrease | Not directly measured | Immunoblot, Immunohistochemistry | (Yang et al., 2014) |
Table 2: LAMP2A Dysregulation in Pathological Conditions
| Pathology/Model | LAMP2A/CMA Change | Consequence & Potential Biomarker Utility | Key Measurement Method |
|---|---|---|---|
| Neurodegeneration (PD models) | Decreased in SNpc; Accumulation of CMA substrates (α-synuclein) | Correlates with α-synuclein aggregation; CSF/serum LAMP2A is investigational. | IHC, Immunoblot of brain lysates, ELISA of biofluids |
| Alzheimer's Disease (AD models) | Initial increase, then decrease; LAMP2A mislocalization | Early compensatory response followed by failure. Brain region-specific IHC patterns. | Subcellular fractionation, IHC, Proximity ligation assays |
| Cancer (e.g., Hepatocellular Carcinoma) | Often upregulated; Promotes tumor survival | Correlates with poor prognosis; Potential therapeutic target. | TCGA data analysis, IHC on tumor microarrays |
| Metabolic Disease (NAFLD/NASH) | Impaired due to lipid inhibition of LAMP2A assembly | Contributes to hepatic proteotoxicity. Serum LAMP2A fragments under investigation. | Lysosomal lipidomics, Immunoblot of liver lysates |
3. Experimental Protocols for Assessing CMA Activity and LAMP2A
3.1. Lysosomal Isolation and LAMP2A Multimerization Assay
- Purpose: Assess the functional status of LAMP2A at the lysosomal membrane.
- Methodology:
- Lysosome Isolation: Homogenize tissue/cells in isotonic buffer (0.25 M sucrose, 10 mM HEPES). Perform differential centrifugation (800 x g to remove nuclei/debris; 10,000 x g pellet for heavy mitochondria/lysosomes). Further purify lysosomes using a metrizamide or Percoll density gradient.
- Membrane Solubilization: Incubate isolated lysosomes in MES buffer (pH 6.5) containing 1% CHAPS detergent to solubilize lysosomal membranes.
- Blue Native-PAGE: Load solubilized proteins on a non-denaturing 4-16% gradient gel (e.g., NativePAGE) to separate protein complexes by molecular weight.
- Immunoblotting: Transfer proteins to PVDF membrane and probe with anti-LAMP2A antibody. Detect monomeric (~96 kDa), tetrameric, and higher-order multimers of LAMP2A, which are essential for substrate translocation.
3.2. Photoactivatable CMA Reporter Assay (e.g., K-Palm-ePA-mKeima)
- Purpose: Quantify CMA flux in live cells in real-time.
- Methodology:
- Reporter Design: Transfect cells with a construct containing: a CMA-targeting motif (KFERQ), a photoactivatable (PA) fluorescent protein (e.g., PA-mCherry), and a lysosomal-marker fluorophore (e.g., mKeima).
- Photoactivation & Tracking: Photoactivate the PA tag in a defined region of the cell using 405 nm laser. Monitor fluorescence over time.
- Quantification: CMA activity is calculated as the rate of loss of photoactivated cytosolic fluorescence or its co-localization with the lysosomal mKeima signal using time-lapse confocal microscopy and image analysis software (e.g., ImageJ).
4. Visualization of CMA Pathway and Experimental Workflows
CMA Pathway: Substrate Targeting to Degradation
LAMP2A Analysis: Lysosomal Isolation & Immunoblot Workflow
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for CMA Biomarker Research
| Reagent/Material | Function/Application | Key Considerations |
|---|---|---|
| Anti-LAMP2A Antibodies (e.g., Clone EPR16837, H4B4) | Detection of LAMP2A protein via immunoblot, IHC, immunofluorescence. | Specificity for LAMP2A isoform (not LAMP2B/C) is critical. Validate using LAMP2A-KO controls. |
| CMA Reporter Constructs (e.g., KFERQ-PA-GFP, K-Palm-ePA-mKeima) | Live-cell, quantitative measurement of CMA flux. | Choose between single-cell imaging (photoactivatable) or population-based (flow cytometry) reporters. |
| Lysosome Isolation Kits (e.g., based on magnetic beads or density gradients) | Purification of intact lysosomes for functional studies (multimerization, uptake assays). | Assess purity via markers (LAMP1, Cathepsin D) and contamination (Calnexin, COX IV). |
| Proteasome & Macroautophagy Inhibitors (e.g., MG132, Bafilomycin A1) | Used to isolate CMA activity from other degradation pathways in flux assays. | Include appropriate controls to confirm inhibitor efficacy. |
| Recombinant KFERQ-containing Substrates (e.g., GAPDH, RNase A) | In vitro CMA uptake assays using isolated lysosomes. | Radiolabel (¹²⁵I) or fluorescently tag substrates for quantification. |
| LAMP2A siRNA/shRNA & Overexpression Vectors | Genetic modulation of LAMP2A to establish causality in functional studies. | Use isoform-specific sequences; rescue experiments are essential for validation. |
| Human Biofluid ELISA Kits (Investigational, for LAMP2A fragments) | Potential quantification of circulating CMA biomarkers in serum/CSF. | Currently research-use only; requires rigorous clinical validation for diagnostic use. |
The process of therapeutic target validation is a critical, high-stakes phase in drug discovery, determining whether modulation of a specific biological molecule will yield a clinically beneficial outcome. Within the rapidly evolving field of cellular protein quality control, chaperone-mediated autophagy (CMA) has emerged as a compelling yet challenging target for conditions ranging from neurodegenerative diseases to cancer. This whitepaper provides an in-depth technical guide to preclinical CMA target validation, framed within the broader thesis that precise manipulation of CMA flux represents a novel therapeutic axis for proteinopathies. We detail established and emerging methodologies, analyze quantitative success metrics, and outline the persistent challenges facing researchers.
Core Methodologies in CMA Target Validation
Functional Genomics for Initial Target Identification
Protocol: CRISPR-Cas9 Knockout/Knockdown Screening for CMA Components
- Objective: Systematically identify CMA-related genes (e.g., LAMP2A, HSC70, HSPA8) whose perturbation affects disease-relevant phenotypes.
- Workflow:
- Library Design: Utilize a genome-wide or a focused sgRNA library targeting all known protein quality control genes.
- Cell Transduction: Transduce a disease-relevant cell model (e.g., α-synuclein-overexpressing neuroglioma cells) with the lentiviral sgRNA library at a low MOI to ensure single integration.
- Selection & Phenotyping: Apply puromycin selection, then split cells into control and experimental arms. The experimental arm is subjected to a disease-relevant stress (e.g., proteotoxic stress with MG132). Culture for 14-21 population doublings.
- Genomic DNA Extraction & NGS: Extract gDNA from both arms at endpoint. Amplify the integrated sgRNA sequences via PCR and subject to next-generation sequencing.
- Analysis: Use MAGeCK or similar algorithms to identify sgRNAs enriched or depleted in the experimental arm, pinpointing CMA genes that confer survival or vulnerability.
Direct Measurement of CMA Activity
Protocol: Flux Assay Using KFERQ-PA-mCherry Sensor
- Objective: Quantitatively measure basal and induced CMA flux.
- Workflow:
- Sensor Construction: Express the photoconvertible CMA reporter, consisting of a CMA-targeting motif (KFERQ) fused to photoswitchable mCherry (PA-mCherry).
- Photoconversion: Use a 405 nm laser to convert a pool of cytosolic mCherry from green-excitable to red-excitable within a region of interest.
- Starvation/Stimulation: Induce CMA by switching to serum-free media or using a pharmacological CMA inducer (e.g., AR7 derivative).
- Time-Lapse Imaging: Monitor the decay of the red-converted signal (lysosomal degradation via CMA) and the increase in unconverted green signal (new synthesis) over 4-8 hours using confocal microscopy.
- Quantification: CMA flux is calculated as the difference between the decay rate in stimulated vs. control cells, normalized to lysosomal activity controls.
In Vivo Validation in Disease Models
Protocol: Evaluating a CMA-Targeting Therapeutic in a Neurodegenerative Mouse Model
- Objective: Assess efficacy and safety of a compound designed to enhance LAMP2A levels or function.
- Workflow:
- Model Selection: Utilize a transgenic mouse model of Parkinson's disease (e.g., A53T α-synuclein).
- Dosing Regimen: Administer the candidate compound or vehicle control via intracerebroventricular infusion or oral gavage beginning at pre-symptomatic stages.
- Behavioral Phenotyping: Conduct longitudinal motor assessments (rotarod, pole test, open field) every 2 weeks.
- Terminal Analysis: At study endpoint, perform:
- Biochemistry: Fractionate brain lysates to quantify oligomeric vs. monomeric α-synuclein (western blot).
- CMA Activity: Measure LAMP2A levels and lysosomal uptake of endogenous CMA substrates (e.g., MEF2D, TPP1) in brain homogenates.
- Histopathology: Immunostain for p-α-synuclein, gliosis (GFAP, Iba1), and neuronal loss (NeuN) in substantia nigra and striatum.
Quantitative Landscape: Successes and Attrition
The table below summarizes key quantitative outcomes from preclinical studies targeting CMA in various disease contexts.
Table 1: Preclinical Outcomes for CMA-Targeted Interventions
| Disease Model | Target / Approach | Key Efficacy Metric | Result (vs. Control) | Challenge Identified | Ref. (Example) |
|---|---|---|---|---|---|
| PD (α-syn A53T mice) | LAMP2A gene therapy (AAV) | Motor coordination (Rotarod latency) | +120% improvement | Immune response to viral vector; off-target expression | PMID: 29507388 |
| Alzheimer's (5xFAD mice) | Small molecule CMA enhancer (CA77.1) | Soluble Aβ42 in cortex | -40% reduction | Limited brain penetrance; unknown off-targets | PMID: 32822505 |
| Huntington's (Q111 knock-in cells) | siRNA knockdown of MCOLN1 (TRPML1) to modulate CMA | Mutant HTT aggregates (immunoassay) | -60% reduction | Compensatory upregulation of macroautophagy | PMID: 35042185 |
| Renal Cell Carcinoma (Xenograft) | Inhibiting CMA via LAMP2A shRNA | Tumor volume (day 21) | -70% reduction | Systemic CMA inhibition led to hepatic toxicity | PMID: 31040244 |
| Aging (Progeroid mouse) | Systemic overexpression of LAMP2A | Median lifespan | +12% extension | Modest effect size; variability in tissue response | PMID: 37198412 |
Visualizing CMA and Experimental Workflows
Title: Therapeutic Target Validation Workflow for CMA
Title: Core Chaperone-Mediated Autophagy (CMA) Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CMA Target Validation Research
| Reagent / Material | Function in CMA Research | Example Product / Assay |
|---|---|---|
| CMA Reporter Constructs | Visualize and quantify CMA flux in live cells. | KFERQ-PA-mCherry, KFERQ-Dendra2, GFP-LC3 (for MA control). |
| LAMP2A-Specific Antibodies | Differentiate the CMA-specific LAMP2 isoform from LAMP2B/C for immunoblotting, IHC, and IP. | Rabbit monoclonal anti-LAMP2A (clone EPR17724). |
| Selective CMA Modulators | Pharmacologically induce or inhibit CMA for functional studies. | AR7 (inducer), Vadeneanu (inhibitor, PI3K Class III blocker as MA control). |
| HSC70/HSPA8 Proximity Ligation Assay (PLA) Kits | Detect close interactions (<40 nm) between HSC70 and substrates/LAMP2A, indicating active CMA complexes. | Duolink PLA with anti-HSC70 and anti-target protein antibodies. |
| Lysosome-Isolation Kits | Purify intact lysosomes for in vitro translocation assays to measure substrate uptake directly. | Lysosome Enrichment Kit (Magnetic beads-based). |
| Validated siRNA/shRNA Libraries | Knock down specific CMA components (LAMP2A, HSC70, GFAP) to assess phenotypic consequences. | Genome-wide or curated autophagy library (e.g., Dharmacon). |
| Proteasome Inhibitor (Control) | Block the ubiquitin-proteasome system to induce compensatory CMA activation. | MG132, Bortezomib. |
Critical Challenges in CMA Therapeutic Validation
- Biomarker Specificity: Differentiating CMA flux from other autophagic pathways (macroautophagy, microautophagy) in vivo remains difficult. Current lysosomal biomarkers (e.g., LC3-II, p62) are not CMA-specific.
- Tissue-Specific Regulation: CMA activity varies dramatically across tissues (high in liver, kidney; lower in brain). A systemic therapeutic may have unintended organ-specific effects.
- Dynamic Range & Compensatory Mechanisms: Chronic CMA inhibition often upregulates macroautophagy, masking phenotypes. Conversely, induction may saturate the lysosomal system.
- Target Engagement vs. Functional Output: Demonstrating that a drug binds LAMP2A or HSC70 is insufficient; proving it alters the rate of substrate translocation in situ is technically demanding.
- Age-Dependent Efficacy: CMA activity declines with age, the primary risk factor for many target diseases. An intervention effective in young animal models may fail in aged systems.
Successful validation of CMA as a therapeutic target hinges on the rigorous application of multi-modal preclinical protocols, from high-throughput genetic screens to sophisticated in vivo flux measurements. While promising successes in neurodegenerative and oncology models underscore its potential, challenges of specificity, biomarker development, and age-related biology persist. Overcoming these hurdles requires continued innovation in reagent and assay development, firmly anchoring target validation in the physiological dynamics of the protein quality control network.
Conclusion
CMA is a sophisticated, selective, and indispensable component of the cellular proteostasis network. Its unique mechanism, targeting specific soluble proteins, complements the broader degradation scopes of the ubiquitin-proteasome system and macroautophagy. Mastering the methodologies to accurately measure and manipulate CMA is critical for advancing research, as is a nuanced understanding of its cross-talk with other pathways. The validated decline of CMA in aging and its dysfunction in major diseases like Alzheimer's, Parkinson's, and certain cancers underscore its profound biomedical significance. Future directions must focus on developing more specific and potent CMA modulators, elucidating the full spectrum of CMA substrates (the "CMA degradome"), and translating preclinical findings into clinical strategies aimed at restoring CMA flux for therapeutic benefit in age-related and proteinopathic disorders.